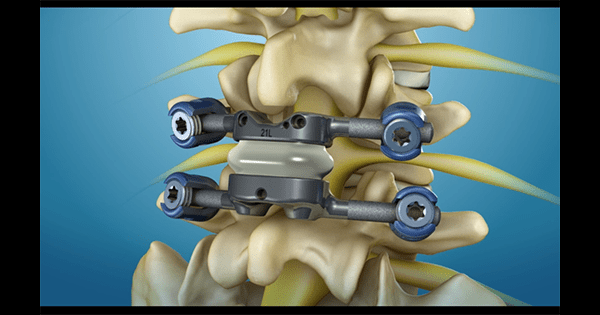
Premia Spine Secures FDA Approval for IDE Study of TOPS
Premia Spine secured FDA approval for a pivotal IDE study of TOPS™, a posterior arthroplasty device for the treatment of spondylolisthesis and spinal stenosis. The study seeks to establish the superiority of TOPS vs. traditional lumbar spinal fusion. May 08, 2017