Copy to clipboard
Copy to clipboard 
Zimmer Biomet was granted FDA 510(k) clearance to market the ROSA Shoulder System for robotic-assisted shoulder replacement surgery. The system is slated for U.S. launch in 2H24.
ROSA Shoulder represents the world’s first robotic surgery system for shoulder replacement, and the fourth application for the company’s ROSA Robotics portfolio, which includes ROSA Knee and ROSA Hip for total joint replacement.
ROSA Shoulder will work with the mymobility Digital Care Management Platform and supports a portfolio that includes the Identity Shoulder. The system joins ZBEdge Dynamic Intelligence, enhancing the company’s digital technologies, robotics and implant solutions.
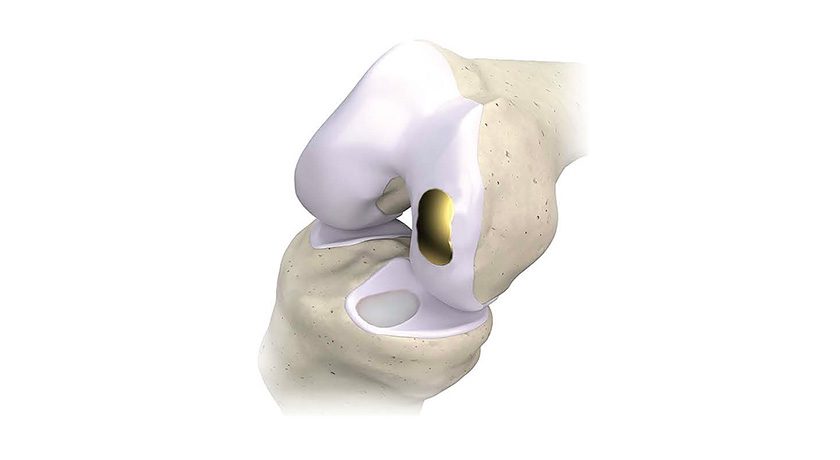
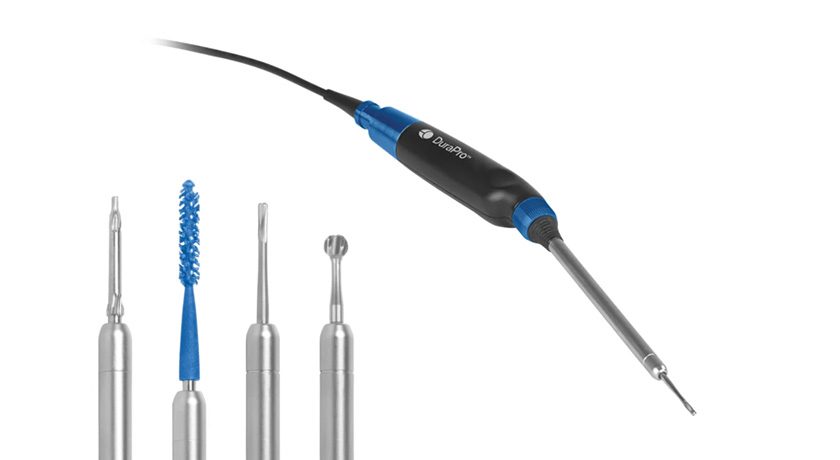
ROSA Shoulder is designed to give surgeons the flexibility to execute a total shoulder replacement using anatomic or reverse techniques and to enable precise placement for improved outcomes. ROSA Shoulder can reproduce humeral head resectioning, and can ease insertion of instruments into incisions by requiring no pin in the center of the glenoid during procedures.
Like all ROSA Robotics applications, ROSA Shoulder is designed to support data-informed physician decision-making based on a patient’s unique anatomy. Pre-operatively, ROSA Shoulder integrates with the newly released Signature ONE Surgical Planning System 2.0, which uses a 3D image-based approach to visualization, surgical planning and patient-specific guide creation. During surgery, the platform provides surgeons with real-time, intra-operative data to help control, execute and validate personalized plans for glenoid and humeral placement, with a goal to reduce complications for patients.
Ivan Tornos, President and Chief Executive Officer at Zimmer Biomet, said, “ROSA Shoulder represents a novel advancement intended to help surgeons leverage the power of robotics and data analytics to perform highly complex shoulder procedures with accuracy and efficiency.”
Source: Zimmer Biomet
Zimmer Biomet was granted FDA 510(k) clearance to market the ROSA Shoulder System for robotic-assisted shoulder replacement surgery. The system is slated for U.S. launch in 2H24.
ROSA Shoulder represents the world’s first robotic surgery system for shoulder replacement, and the fourth application for the company’s ROSA Robotics portfolio,...
Zimmer Biomet was granted FDA 510(k) clearance to market the ROSA Shoulder System for robotic-assisted shoulder replacement surgery. The system is slated for U.S. launch in 2H24.
ROSA Shoulder represents the world’s first robotic surgery system for shoulder replacement, and the fourth application for the company’s ROSA Robotics portfolio, which includes ROSA Knee and ROSA Hip for total joint replacement.
ROSA Shoulder will work with the mymobility Digital Care Management Platform and supports a portfolio that includes the Identity Shoulder. The system joins ZBEdge Dynamic Intelligence, enhancing the company’s digital technologies, robotics and implant solutions.
ROSA Shoulder is designed to give surgeons the flexibility to execute a total shoulder replacement using anatomic or reverse techniques and to enable precise placement for improved outcomes. ROSA Shoulder can reproduce humeral head resectioning, and can ease insertion of instruments into incisions by requiring no pin in the center of the glenoid during procedures.
Like all ROSA Robotics applications, ROSA Shoulder is designed to support data-informed physician decision-making based on a patient’s unique anatomy. Pre-operatively, ROSA Shoulder integrates with the newly released Signature ONE Surgical Planning System 2.0, which uses a 3D image-based approach to visualization, surgical planning and patient-specific guide creation. During surgery, the platform provides surgeons with real-time, intra-operative data to help control, execute and validate personalized plans for glenoid and humeral placement, with a goal to reduce complications for patients.
Ivan Tornos, President and Chief Executive Officer at Zimmer Biomet, said, “ROSA Shoulder represents a novel advancement intended to help surgeons leverage the power of robotics and data analytics to perform highly complex shoulder procedures with accuracy and efficiency.”
Source: Zimmer Biomet

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.