
 Copy to clipboard
Copy to clipboard 
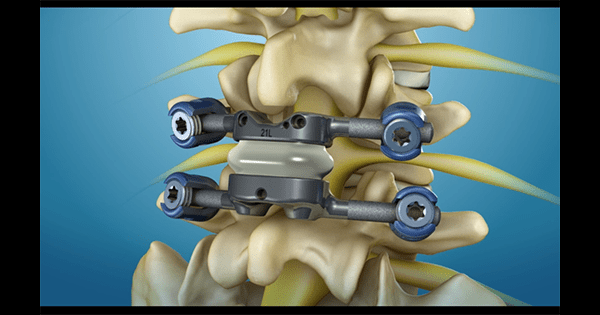
The TOPS™ facet arthroplasty system from Premia Spine received Breakthrough Device Designation from FDA. TOPS is the first and only facet joint replacement system for the lumbar spine.
Developed to treat spinal stenosis or spondylolisthesis, the system is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability and durability after decompression. TOPS is currently the subject of a pivotal clinical trial under an Investigational Device Exemption from FDA.
“The TOPS System is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability, and durability for patients,” said Premia Spine CEO Ron Sacher. “We are gratified that the FDA recognizes TOPS’s potential to advance the standard of care for patients with spinal stenosis and spondylolisthesis.”
The TOPS™ facet arthroplasty system from Premia Spine received Breakthrough Device Designation from FDA. TOPS is the first and only facet joint replacement system for the lumbar spine.
Developed to treat spinal stenosis or spondylolisthesis, the system is designed to relieve debilitating lower back and leg pain while enabling long-term...
The TOPS™ facet arthroplasty system from Premia Spine received Breakthrough Device Designation from FDA. TOPS is the first and only facet joint replacement system for the lumbar spine.
Developed to treat spinal stenosis or spondylolisthesis, the system is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability and durability after decompression. TOPS is currently the subject of a pivotal clinical trial under an Investigational Device Exemption from FDA.
“The TOPS System is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability, and durability for patients,” said Premia Spine CEO Ron Sacher. “We are gratified that the FDA recognizes TOPS’s potential to advance the standard of care for patients with spinal stenosis and spondylolisthesis.”

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







