
 Copy to clipboard
Copy to clipboard 
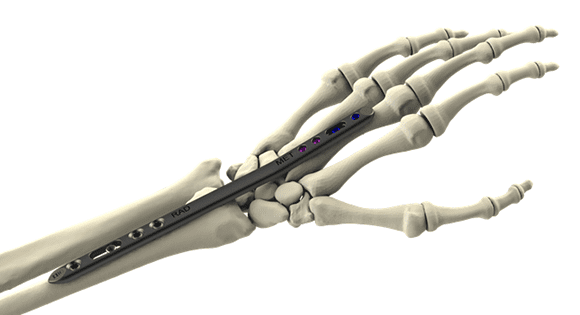
The Orthopaedic Implant Company (OIC) announced the FDA clearance and commercial launch of what the company calls the market’s first and only high-value dorsal spanning plate. This addition to the company’s orthopedic trauma implant portfolio expands the potential clinical impact of OIC’s DRPx wrist fracture plating system.
Launched in 2021, DRPx is the only high-value distal radius plating system available in the U.S., supporting a wide array of orthopedic surgeons’ technique preferences while increasing opportunities for cost savings to help improve the financial viability of ambulatory surgery centers, hospitals and their patients nationwide.
OIC’s wrist fracture plating system reportedly reduces implant costs, on average, by more than 50% and provides biomechanical and clinical outcome equivalency in comparison to branded and premium-priced implant systems.
Itai Nemovicher, President and CEO, said, “The reality right now is that ASCs are only being reimbursed at 53% the rate we’re seeing with hospitals performing the exact same procedures, so this is where we can make a huge difference for ASCs. Adding the dorsal spanning plate to our DRPx system provides surgeons and facilities with the chance to substantially increase value on a high-volume procedure, streamline operations and get the exact same clinical results they’re getting with more expensive implant systems.”
DRPx is commercially available in the United States.
Source: The Orthopaedic Implant Company
The Orthopaedic Implant Company (OIC) announced the FDA clearance and commercial launch of what the company calls the market's first and only high-value dorsal spanning plate. This addition to the company's orthopedic trauma implant portfolio expands the potential clinical impact of OIC's DRPx wrist fracture plating system.
Launched in 2021,...
The Orthopaedic Implant Company (OIC) announced the FDA clearance and commercial launch of what the company calls the market’s first and only high-value dorsal spanning plate. This addition to the company’s orthopedic trauma implant portfolio expands the potential clinical impact of OIC’s DRPx wrist fracture plating system.
Launched in 2021, DRPx is the only high-value distal radius plating system available in the U.S., supporting a wide array of orthopedic surgeons’ technique preferences while increasing opportunities for cost savings to help improve the financial viability of ambulatory surgery centers, hospitals and their patients nationwide.
OIC’s wrist fracture plating system reportedly reduces implant costs, on average, by more than 50% and provides biomechanical and clinical outcome equivalency in comparison to branded and premium-priced implant systems.
Itai Nemovicher, President and CEO, said, “The reality right now is that ASCs are only being reimbursed at 53% the rate we’re seeing with hospitals performing the exact same procedures, so this is where we can make a huge difference for ASCs. Adding the dorsal spanning plate to our DRPx system provides surgeons and facilities with the chance to substantially increase value on a high-volume procedure, streamline operations and get the exact same clinical results they’re getting with more expensive implant systems.”
DRPx is commercially available in the United States.
Source: The Orthopaedic Implant Company

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







