
 Copy to clipboard
Copy to clipboard 
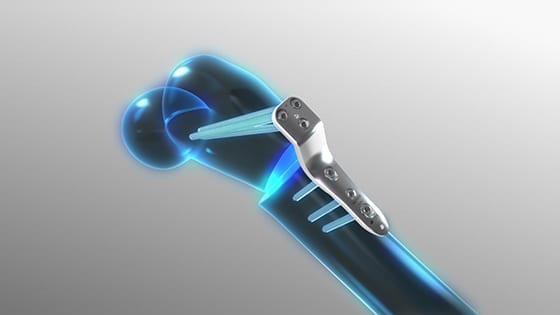
Orthofix Medical received CE Mark approval and FDA 510(k) clearance to market the JuniOrtho® Plating System. Created specifically for pediatric patients, JuniOrtho is designed to support advanced deformity and trauma reconstruction of the lower extremities.
The system is complemented by pre-operative planning software that can streamline implant selection. Specifically developed for use with JuniOrtho Plating, the software is currently available in Europe and planned for release in the U.S. later this year.
Offered in a range of plate sizes with a variety of lengths, JuniOrtho accommodates locking and non-locking screws and comprises sterile implants and single-use tools to reduce the risk of contamination and optimize procedure efficiency.
JuniOrtho Plating is part of the JuniOrtho line of pediatric solutions that includes the TL-HEX™, TrueLok Ring Fixation, eight-Plate Guided Growth System+, etc. The JuniOrtho brand launched in 2017.
“The JuniOrtho Plating System represents our continued commitment to advancing pediatric orthopedics by providing surgeons the devices they need to treat even the smallest of patients,” said Jon Serbousek, President and CEO. “We are excited to now be able to offer surgeons both an internal and external fixation systems to expand our portfolio of pediatric deformity care solutions.”
Orthofix Medical received CE Mark approval and FDA 510(k) clearance to market the JuniOrtho® Plating System. Created specifically for pediatric patients, JuniOrtho is designed to support advanced deformity and trauma reconstruction of the lower extremities.
The system is complemented by pre-operative planning software that can streamline...
Orthofix Medical received CE Mark approval and FDA 510(k) clearance to market the JuniOrtho® Plating System. Created specifically for pediatric patients, JuniOrtho is designed to support advanced deformity and trauma reconstruction of the lower extremities.
The system is complemented by pre-operative planning software that can streamline implant selection. Specifically developed for use with JuniOrtho Plating, the software is currently available in Europe and planned for release in the U.S. later this year.
Offered in a range of plate sizes with a variety of lengths, JuniOrtho accommodates locking and non-locking screws and comprises sterile implants and single-use tools to reduce the risk of contamination and optimize procedure efficiency.
JuniOrtho Plating is part of the JuniOrtho line of pediatric solutions that includes the TL-HEX™, TrueLok Ring Fixation, eight-Plate Guided Growth System+, etc. The JuniOrtho brand launched in 2017.
“The JuniOrtho Plating System represents our continued commitment to advancing pediatric orthopedics by providing surgeons the devices they need to treat even the smallest of patients,” said Jon Serbousek, President and CEO. “We are excited to now be able to offer surgeons both an internal and external fixation systems to expand our portfolio of pediatric deformity care solutions.”

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







