
 Copy to clipboard
Copy to clipboard 
Ortoma signed an agreement with Johnson & Johnson K.K. for exclusive distribution rights in Japan of the Ortoma Treatment Solution for hip surgery. It is the first agreement that the company has executed with a global strategic partner, and the agreement allows for the possibility to expand the partnership to other global markets.
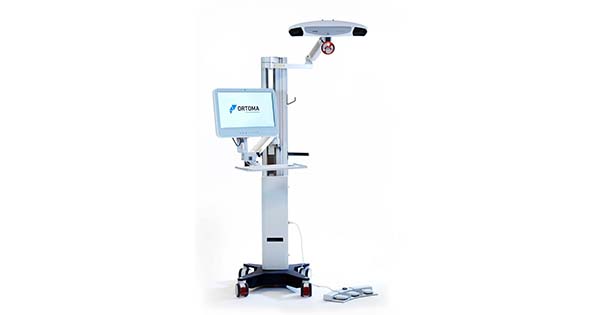
Ortoma is the provider of the Ortoma Treatment Solution (OTS), which includes OTS Hip Plan and OTS Hip Guide for pre-operative planning, intra-operative navigation and post-operative follow-up of hip implant surgical procedures. The OTS platform is assisted by Artificial Intelligence for improved accuracy, efficiency and surgical outcomes.
OTS Hip Plan can be used with Depuy Synthes hip implants available on the Japanese market. The hip plan module obtained certification in Japan for pre-operative planning of hip replacement earlier this month.
The agreement includes the commercial terms for the exclusive distribution rights in Japan of OTS for hip replacement surgery including the products OTS Hip Plan and OTS Hip Guide. The agreement is valid for an initial term of seven years. Johnson & Johnson K.K.with affiliated companies has the right to negotiate an extension of the strategic partnership to other surgical areas in Japan as well as to other global markets.
Johnson & Johnson K.K. will evaluate the products clinically during an initial pilot period in 2022 agreed upon by the parties. If satisfactory, Johnson & Johnson K.K.will proceed with the launch of the products on the Japanese market. The evaluation will be conducted following the registration of appropriate regulatory approval of the relevant OTS product in Japan. OTS Hip Plan already has regulatory approval in Japan. The Johnson & Johnson K.K. has the right to terminate the agreement two years after regulatory approval.
Source: Ortoma
Ortoma signed an agreement with Johnson & Johnson K.K. for exclusive distribution rights in Japan of the Ortoma Treatment Solution for hip surgery. It is the first agreement that the company has executed with a global strategic partner, and the agreement allows for the possibility to expand the partnership to other global markets.
Ortoma is...
Ortoma signed an agreement with Johnson & Johnson K.K. for exclusive distribution rights in Japan of the Ortoma Treatment Solution for hip surgery. It is the first agreement that the company has executed with a global strategic partner, and the agreement allows for the possibility to expand the partnership to other global markets.
Ortoma is the provider of the Ortoma Treatment Solution (OTS), which includes OTS Hip Plan and OTS Hip Guide for pre-operative planning, intra-operative navigation and post-operative follow-up of hip implant surgical procedures. The OTS platform is assisted by Artificial Intelligence for improved accuracy, efficiency and surgical outcomes.
OTS Hip Plan can be used with Depuy Synthes hip implants available on the Japanese market. The hip plan module obtained certification in Japan for pre-operative planning of hip replacement earlier this month.
The agreement includes the commercial terms for the exclusive distribution rights in Japan of OTS for hip replacement surgery including the products OTS Hip Plan and OTS Hip Guide. The agreement is valid for an initial term of seven years. Johnson & Johnson K.K.with affiliated companies has the right to negotiate an extension of the strategic partnership to other surgical areas in Japan as well as to other global markets.
Johnson & Johnson K.K. will evaluate the products clinically during an initial pilot period in 2022 agreed upon by the parties. If satisfactory, Johnson & Johnson K.K.will proceed with the launch of the products on the Japanese market. The evaluation will be conducted following the registration of appropriate regulatory approval of the relevant OTS product in Japan. OTS Hip Plan already has regulatory approval in Japan. The Johnson & Johnson K.K. has the right to terminate the agreement two years after regulatory approval.
Source: Ortoma

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







