
 Copy to clipboard
Copy to clipboard 
CartiHeal announced the first commercial implantation of Agili-C in the U.S.
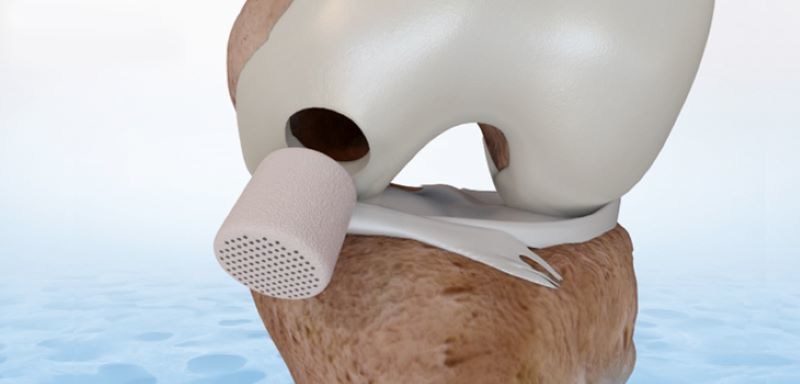
The Agili-C implant features a bi-phasic design that supports both bone remodeling and cartilage regeneration. Agili-C received FDA breakthrough designation status in 2020 and demonstrated clinical superiority to the surgical standard of care (microfracture and debridement) in one of the largest cartilage clinical trials on record with the broadest inclusion criteria, which was meant to mimic real-life patients. This resulted in the Agili-C implant receiving FDA approval with an indication for the treatment of an International Cartilage Repair Society (ICRS) grade III or above knee-joint surface lesion(s).
“We are thrilled to share the Agili-C technology with the surgical community with a goal for it to reach every surgical facility in the US and beyond,” said Nir Altschuler, CartiHeal’s Founder and CEO. “This is a milestone achievement for everyone at CartiHeal and we would like to congratulate Dr. Zaslav on successfully performing our first commercial case in the U.S.”
Agili-C is available throughout the U.S.
Source: CartiHeal
CartiHeal announced the first commercial implantation of Agili-C in the U.S.
The Agili-C implant features a bi-phasic design that supports both bone remodeling and cartilage regeneration. Agili-C received FDA breakthrough designation status in 2020 and demonstrated clinical superiority to the surgical standard of care (microfracture and...
CartiHeal announced the first commercial implantation of Agili-C in the U.S.
The Agili-C implant features a bi-phasic design that supports both bone remodeling and cartilage regeneration. Agili-C received FDA breakthrough designation status in 2020 and demonstrated clinical superiority to the surgical standard of care (microfracture and debridement) in one of the largest cartilage clinical trials on record with the broadest inclusion criteria, which was meant to mimic real-life patients. This resulted in the Agili-C implant receiving FDA approval with an indication for the treatment of an International Cartilage Repair Society (ICRS) grade III or above knee-joint surface lesion(s).
“We are thrilled to share the Agili-C technology with the surgical community with a goal for it to reach every surgical facility in the US and beyond,” said Nir Altschuler, CartiHeal’s Founder and CEO. “This is a milestone achievement for everyone at CartiHeal and we would like to congratulate Dr. Zaslav on successfully performing our first commercial case in the U.S.”
Agili-C is available throughout the U.S.
Source: CartiHeal

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







