Episurf Medical is planning the 500th surgery with the Episealer® implant, which will occur in Germany.
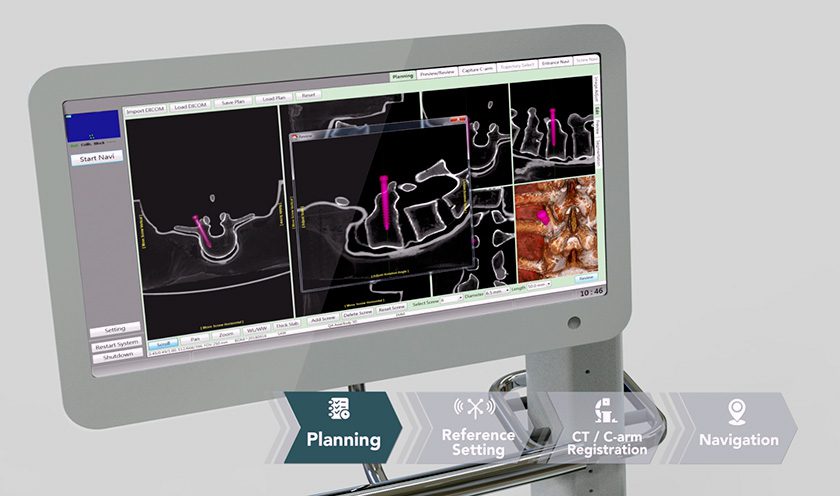
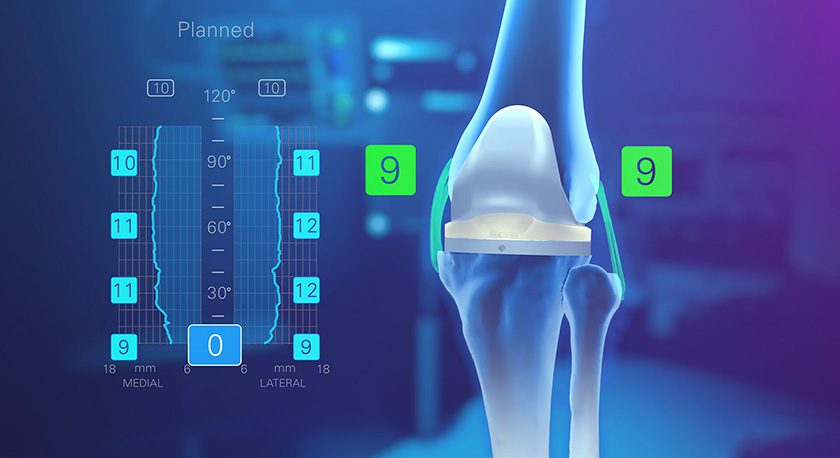
Episealer devices treat localized cartilage damage in the knee. The implants and associated surgical tools are approved under the CE Mark. At the end of 2018, the company announced FDA approval for its Investigational Device Exemption application to initiate a clinical trial of the Episealer knee implant; the two-year prospective, randomized, controlled, multi-center study will evaluate the device vs. microfracture in the treatment of focal femoral knee chondral or osteochondral lesions in 180 patients in the U.S. and Europe.
“Reaching this milestone while we are experiencing a continuous flow of successful patient outcomes is of great importance to us. Our customers are constantly talking to us about the challenge in treating the middle-aged patients who suffer from early cartilage and bone lesions in the knee joint. It is our vision to help these patients to return to a normal lifestyle, and we are very determined on delivering on this vision,” says Pål Ryfors, CEO Episurf Medical.
Source: Episurf Medical AB
Episurf Medical is planning the 500th surgery with the Episealer® implant, which will occur in Germany.
Episealer devices treat localized cartilage damage in the knee. The implants and associated surgical tools are approved under the CE Mark. At the end of 2018, the company announced FDA approval for its Investigational Device Exemption...
Episurf Medical is planning the 500th surgery with the Episealer® implant, which will occur in Germany.
Episealer devices treat localized cartilage damage in the knee. The implants and associated surgical tools are approved under the CE Mark. At the end of 2018, the company announced FDA approval for its Investigational Device Exemption application to initiate a clinical trial of the Episealer knee implant; the two-year prospective, randomized, controlled, multi-center study will evaluate the device vs. microfracture in the treatment of focal femoral knee chondral or osteochondral lesions in 180 patients in the U.S. and Europe.
“Reaching this milestone while we are experiencing a continuous flow of successful patient outcomes is of great importance to us. Our customers are constantly talking to us about the challenge in treating the middle-aged patients who suffer from early cartilage and bone lesions in the knee joint. It is our vision to help these patients to return to a normal lifestyle, and we are very determined on delivering on this vision,” says Pål Ryfors, CEO Episurf Medical.
Source: Episurf Medical AB


You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.