
 Copy to clipboard
Copy to clipboard 
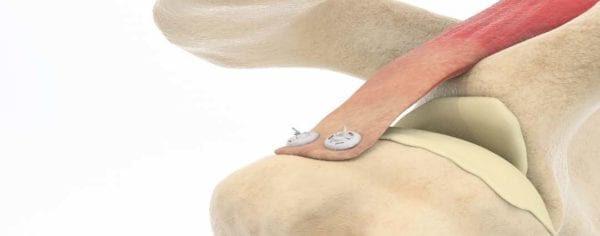
Ziptek is introducing the second generation of the ZipE® resorbable locking suture button kits.
Since its FDA 510(k) clearance, the device has been employed in over 40 arthroscopic rotator cuff repairs, some with one-year follow-up. The device has FDA cleared Indications for Use in five major joints and 24 specific surgical indications, and to date ZipE has been implanted for medial collateral ligament and triceps avulsion repair, open subscapularis repair of the rotator cuff, subscapularis closure following a total shoulder replacement and Haglund Achilles reattachment.
ZipE is deployed like a zip-tie and is designed to create a larger tissue-to-bone contact area, reducing the potential for gap formation and providing a stronger construct to resist rip-through. The suture fixation technology resorbs after three months.
Source: Ziptek LLC
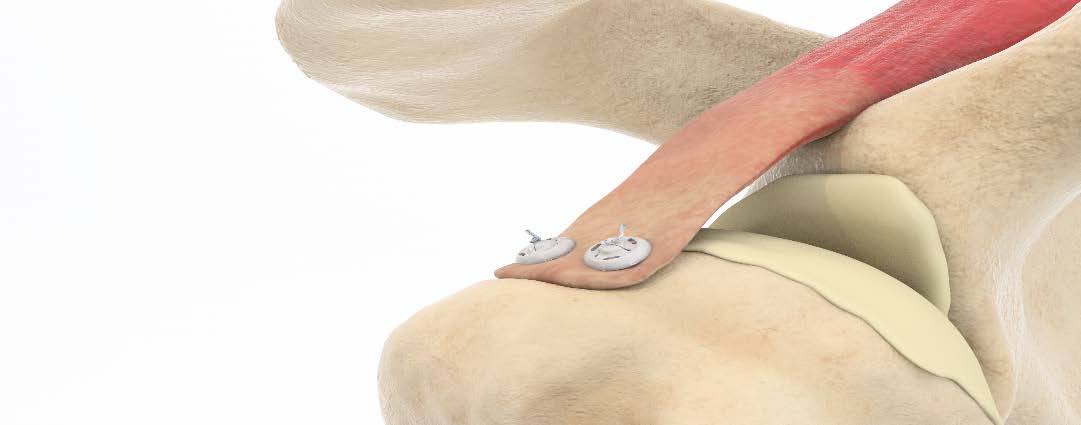
Image courtesy of Ziptek LLC
Ziptek is introducing the second generation of the ZipE® resorbable locking suture button kits.
Since its FDA 510(k) clearance, the device has been employed in over 40 arthroscopic rotator cuff repairs, some with one-year follow-up. The device has FDA cleared Indications for Use in five major joints and 24 specific surgical indications, and to...
Ziptek is introducing the second generation of the ZipE® resorbable locking suture button kits.
Since its FDA 510(k) clearance, the device has been employed in over 40 arthroscopic rotator cuff repairs, some with one-year follow-up. The device has FDA cleared Indications for Use in five major joints and 24 specific surgical indications, and to date ZipE has been implanted for medial collateral ligament and triceps avulsion repair, open subscapularis repair of the rotator cuff, subscapularis closure following a total shoulder replacement and Haglund Achilles reattachment.
ZipE is deployed like a zip-tie and is designed to create a larger tissue-to-bone contact area, reducing the potential for gap formation and providing a stronger construct to resist rip-through. The suture fixation technology resorbs after three months.
Source: Ziptek LLC

Image courtesy of Ziptek LLC

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







