Copy to clipboard
Copy to clipboard 
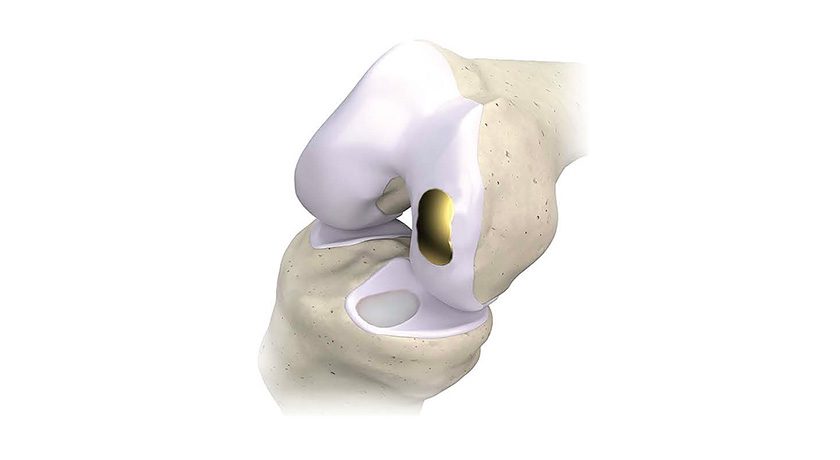
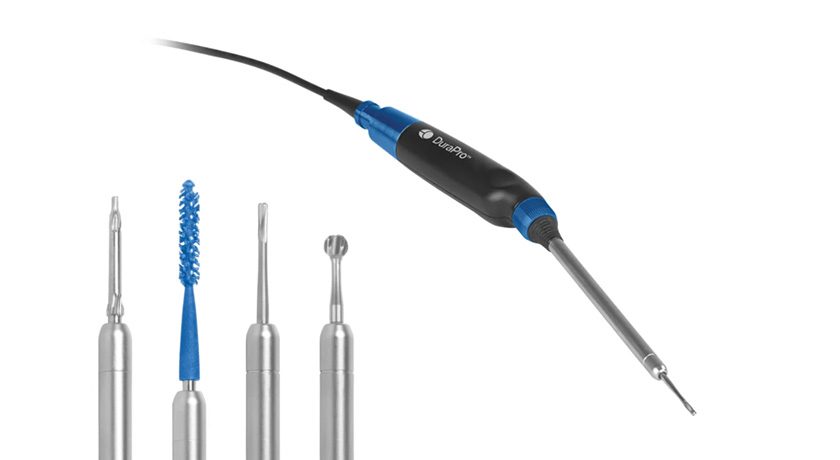
Zavation Medical Products was granted FDA 510(k) clearance to market eZspand™ Lateral, an expandable lumbar interbody fusion device.
eZspand Lateral features expandable precision paired with continual expansion to support an optimized fit for each patient. eZspand Lateral does not require secondary locking, minimizing procedural steps. The cage has up to 15° lordosis and the ability to backfill with bone graft. Each implant expands to 6mm from the insertion height to create a large graft window that facilitates fusion.
Zavation eZspand Interbody System implants are indicated for spinal fusion procedures to be used with autogenous bone graft in skeletally mature patients. eZspand Interbody System implants are intended for use at either one level or two contiguous levels in the lumbar spine, from L2 to S1, for the treatment of degenerative disc disease with up to Grade I spondylolisthesis.
The Zavation eZspand Interbody System implants are intended to be used with supplemental internal fixation appropriate for the implanted level, including the Zavation Spinal System.
Source: Zavation Medical Products
Zavation Medical Products was granted FDA 510(k) clearance to market eZspand™ Lateral, an expandable lumbar interbody fusion device.
eZspand Lateral features expandable precision paired with continual expansion to support an optimized fit for each patient. eZspand Lateral does not require secondary locking, minimizing procedural steps. The cage...
Zavation Medical Products was granted FDA 510(k) clearance to market eZspand™ Lateral, an expandable lumbar interbody fusion device.
eZspand Lateral features expandable precision paired with continual expansion to support an optimized fit for each patient. eZspand Lateral does not require secondary locking, minimizing procedural steps. The cage has up to 15° lordosis and the ability to backfill with bone graft. Each implant expands to 6mm from the insertion height to create a large graft window that facilitates fusion.
Zavation eZspand Interbody System implants are indicated for spinal fusion procedures to be used with autogenous bone graft in skeletally mature patients. eZspand Interbody System implants are intended for use at either one level or two contiguous levels in the lumbar spine, from L2 to S1, for the treatment of degenerative disc disease with up to Grade I spondylolisthesis.
The Zavation eZspand Interbody System implants are intended to be used with supplemental internal fixation appropriate for the implanted level, including the Zavation Spinal System.
Source: Zavation Medical Products

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.