
 Copy to clipboard
Copy to clipboard 
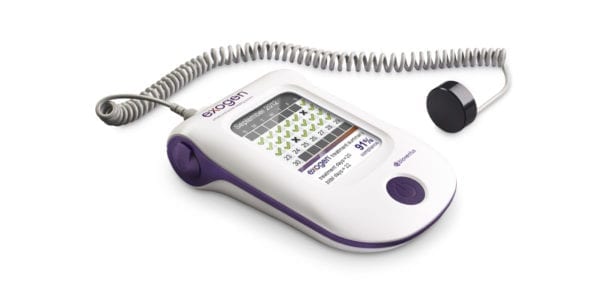
 The U.K.’s National Institute for Health and Clinical Excellence (NICE) released guidance in support of Bioventus’ EXOGEN® ultrasound bone healing device as an alternative to surgery for the treatment of nonunion fractures. NICE originally made its recommendation for EXOGEN in 2013.
The U.K.’s National Institute for Health and Clinical Excellence (NICE) released guidance in support of Bioventus’ EXOGEN® ultrasound bone healing device as an alternative to surgery for the treatment of nonunion fractures. NICE originally made its recommendation for EXOGEN in 2013.
External assessment found that while the cost of revision surgery has increased 25%, costs related to the use of EXOGEN to treat long bone fractures with nonunion has remained the same since previous review in 2013. This has resulted in the doubling of cost savings to £2,407 (~USD $3,000, previously £1,164/$1,400) per patient by avoiding surgery.
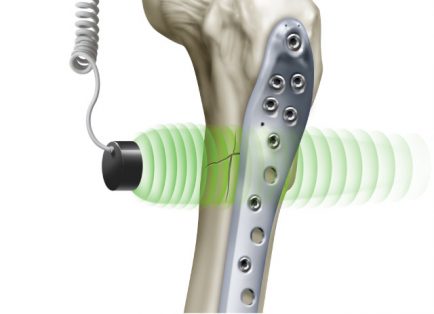
EXOGEN uses low-intensity pulsed ultrasound to stimulate the natural bone healing process and promote fracture healing. It has demonstrated an 86% heal rate for fractures that have not healed on their own, and may provide 38% faster healing of fresh fractures.
Source: Bioventus
The U.K.'s National Institute for Health and Clinical Excellence (NICE) released guidance in support of Bioventus' EXOGEN® ultrasound bone healing device as an alternative to surgery for the treatment of nonunion fractures. NICE originally made its recommendation for EXOGEN in 2013.
External assessment found that while the cost of revision...
 The U.K.’s National Institute for Health and Clinical Excellence (NICE) released guidance in support of Bioventus’ EXOGEN® ultrasound bone healing device as an alternative to surgery for the treatment of nonunion fractures. NICE originally made its recommendation for EXOGEN in 2013.
The U.K.’s National Institute for Health and Clinical Excellence (NICE) released guidance in support of Bioventus’ EXOGEN® ultrasound bone healing device as an alternative to surgery for the treatment of nonunion fractures. NICE originally made its recommendation for EXOGEN in 2013.
External assessment found that while the cost of revision surgery has increased 25%, costs related to the use of EXOGEN to treat long bone fractures with nonunion has remained the same since previous review in 2013. This has resulted in the doubling of cost savings to £2,407 (~USD $3,000, previously £1,164/$1,400) per patient by avoiding surgery.
EXOGEN uses low-intensity pulsed ultrasound to stimulate the natural bone healing process and promote fracture healing. It has demonstrated an 86% heal rate for fractures that have not healed on their own, and may provide 38% faster healing of fresh fractures.
Source: Bioventus

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







