
 Copy to clipboard
Copy to clipboard 
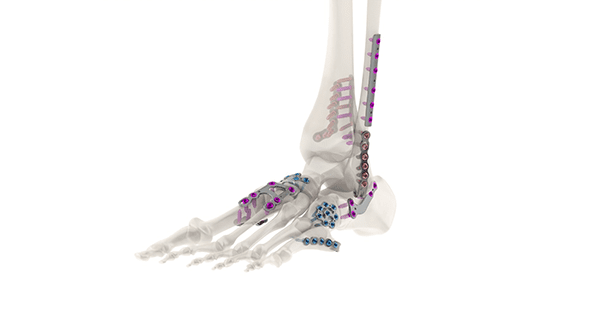
Tyber Medical received its second and largest FDA 510(k) clearance for its expanded line of anatomical plating systems. With more than 1,500 SKUs included in the clearance, Tyber Medical now offers its most comprehensive portfolio of fixation devices for the extremities. The line includes over 73 different indication-specific anatomical plating families.
The indication categories for this most recent clearance include Mini-frag/Small Bone, Long Bone Fracture and Ankle Fracture/Fusion plates. Tyber Medical’s Mini-frag/Small Bone plates incorporate a series of stainless steel and titanium standard and variable locking compression plates and screws of varying lengths, thicknesses, and configurations. This system addresses the stabilization of fractures, osteotomies, joint fusions, non-unions, and fusions of small bones and small bone fragments to the hand, wrist, foot, and ankle. The foot and ankle plating line is used in the fixation of medium/large bones, multi-fragment containment, as well as reconstruction and revision surgeries.
Tyber Medical’s Long Bone Fracture plates comprise a straight, low-contact locking plate and a 1/3 locking tubular plate. These plates incorporate hybrid cortical/cancellous thread featuring both variable angle locking and standard non-locking screws. It is intended for osteotomies and non-unions and the fixation of fractures of the clavicle, scapula, olecranon, humerus, radius, ulna and fibula. The Tyber Medical Ankle Fracture/Fusion System is designed to address a variety of indications in ankle reconstruction mid-shaft and distal tibia/fibula fixation surgery, with a variety of locking plate types for various fibula and tibia positions. Indications for this system include fixation of fractures of the distal tibia, including ankle fractures, intra-, and extra-articular fractures, osteotomies, medial malleolar fractures, and non-unions of the metaphyseal and diaphyseal region of the distal fibula and calcaneus; and In distal tibia/fibula long bones.
The titanium system features color anodized for easy identification of sizing and alignment with color-coded instrumentation. Tyber Medical plans to launch its new fixation systems in 2022, while actively pursuing a CE Mark, utilizing the company’s global clinical registries to collect Post Market Clinical Follow-up (PMCF).
Source: Tyber Medical
Tyber Medical received its second and largest FDA 510(k) clearance for its expanded line of anatomical plating systems. With more than 1,500 SKUs included in the clearance, Tyber Medical now offers its most comprehensive portfolio of fixation devices for the extremities. The line includes over 73 different indication-specific anatomical plating...
Tyber Medical received its second and largest FDA 510(k) clearance for its expanded line of anatomical plating systems. With more than 1,500 SKUs included in the clearance, Tyber Medical now offers its most comprehensive portfolio of fixation devices for the extremities. The line includes over 73 different indication-specific anatomical plating families.
The indication categories for this most recent clearance include Mini-frag/Small Bone, Long Bone Fracture and Ankle Fracture/Fusion plates. Tyber Medical’s Mini-frag/Small Bone plates incorporate a series of stainless steel and titanium standard and variable locking compression plates and screws of varying lengths, thicknesses, and configurations. This system addresses the stabilization of fractures, osteotomies, joint fusions, non-unions, and fusions of small bones and small bone fragments to the hand, wrist, foot, and ankle. The foot and ankle plating line is used in the fixation of medium/large bones, multi-fragment containment, as well as reconstruction and revision surgeries.
Tyber Medical’s Long Bone Fracture plates comprise a straight, low-contact locking plate and a 1/3 locking tubular plate. These plates incorporate hybrid cortical/cancellous thread featuring both variable angle locking and standard non-locking screws. It is intended for osteotomies and non-unions and the fixation of fractures of the clavicle, scapula, olecranon, humerus, radius, ulna and fibula. The Tyber Medical Ankle Fracture/Fusion System is designed to address a variety of indications in ankle reconstruction mid-shaft and distal tibia/fibula fixation surgery, with a variety of locking plate types for various fibula and tibia positions. Indications for this system include fixation of fractures of the distal tibia, including ankle fractures, intra-, and extra-articular fractures, osteotomies, medial malleolar fractures, and non-unions of the metaphyseal and diaphyseal region of the distal fibula and calcaneus; and In distal tibia/fibula long bones.
The titanium system features color anodized for easy identification of sizing and alignment with color-coded instrumentation. Tyber Medical plans to launch its new fixation systems in 2022, while actively pursuing a CE Mark, utilizing the company’s global clinical registries to collect Post Market Clinical Follow-up (PMCF).
Source: Tyber Medical

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







