Copy to clipboard
Copy to clipboard 
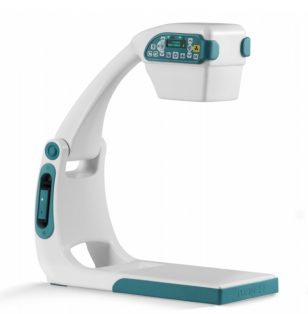 Turner Imaging Systems received FDA 510(k) clearance to market the Smart-C Mini-C Arm Portable Fluoroscopy and X-Ray Imaging Device. Initial U.S. launch will be followed by a global product release.
Turner Imaging Systems received FDA 510(k) clearance to market the Smart-C Mini-C Arm Portable Fluoroscopy and X-Ray Imaging Device. Initial U.S. launch will be followed by a global product release.
Smart-C was developed to support extremity x-rays at the point of care. At only 16 pounds, the battery-powered, cordless device can be easily hand-carried to its destination. It features a collapsible design, as well as an optional stand; an independent articulating arm can be detached for independent tabletop use. This is the first device in Turner’s pipeline that will employ similar technologies as the Smart-C.
“The Smart-C will revolutionize how and where doctors use X-rays,” said D. Clark Turner, Ph.D., founder and CEO of Turner Imaging Systems. “We envision the Smart-C being particularly useful for humanitarian aid workers, including groups such as Doctors Without Borders. Other specialty applications will include imaging in sports medicine, especially on the field or in the locker room, in-office outpatient orthopedic surgeries, care on military battlefields, extremity injections for pain management, mobile radiology units in rural areas, emergency rooms and much more.”
Source: Turner Imaging Systems
Turner Imaging Systems received FDA 510(k) clearance to market the Smart-C Mini-C Arm Portable Fluoroscopy and X-Ray Imaging Device. Initial U.S. launch will be followed by a global product release.
Smart-C was developed to support extremity x-rays at the point of care. At only 16 pounds, the battery-powered, cordless device can be easily...
 Turner Imaging Systems received FDA 510(k) clearance to market the Smart-C Mini-C Arm Portable Fluoroscopy and X-Ray Imaging Device. Initial U.S. launch will be followed by a global product release.
Turner Imaging Systems received FDA 510(k) clearance to market the Smart-C Mini-C Arm Portable Fluoroscopy and X-Ray Imaging Device. Initial U.S. launch will be followed by a global product release.
Smart-C was developed to support extremity x-rays at the point of care. At only 16 pounds, the battery-powered, cordless device can be easily hand-carried to its destination. It features a collapsible design, as well as an optional stand; an independent articulating arm can be detached for independent tabletop use. This is the first device in Turner’s pipeline that will employ similar technologies as the Smart-C.
“The Smart-C will revolutionize how and where doctors use X-rays,” said D. Clark Turner, Ph.D., founder and CEO of Turner Imaging Systems. “We envision the Smart-C being particularly useful for humanitarian aid workers, including groups such as Doctors Without Borders. Other specialty applications will include imaging in sports medicine, especially on the field or in the locker room, in-office outpatient orthopedic surgeries, care on military battlefields, extremity injections for pain management, mobile radiology units in rural areas, emergency rooms and much more.”
Source: Turner Imaging Systems

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.