
 Copy to clipboard
Copy to clipboard 
THINK Surgical entered the South Korean market and completed >10 total knee arthroplasty (TKA) cases using the TSolution One robotic surgical system.
TSolution One received clearance from Korea’s Ministry of Food and Drug Safety in 1Q17 for Total Hip Arthroplasty (THA) and TKA. The THA application is FDA 510(k)-cleared and CE Mark-approved, and plans are in place to obtain these regulatory clearances for total knee. Worldwide, TSolutions’s technology has been used in thousands of hip and knee cases.
The open-platform system uses CT scans to support 3D pre-surgical planning and offers an alternative to manual template planning, broaching, reaming and impacting techniques for preparation of the femoral canal and acetabular cup. It comprises the TPLAN® Preoperative Planning Workstation and TCAT® robotic system: an electromechanical arm, control electronics and computer, display monitor, cutters, drapes, irrigation sets, etc.
Source: THINK Surgical, Inc.
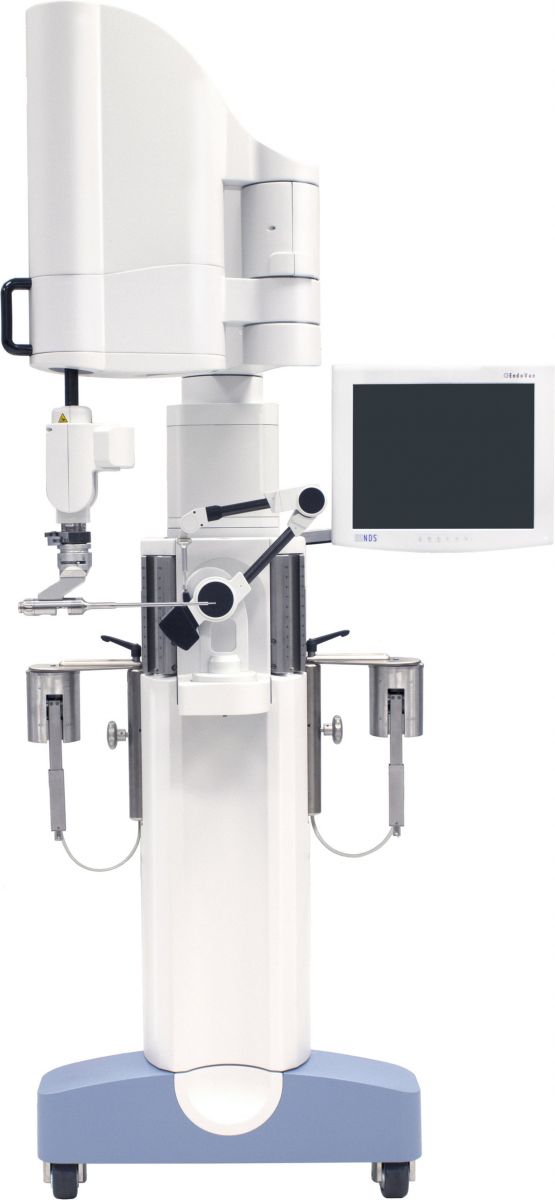
Image courtesy of THINK Surgical, Inc.
THINK Surgical entered the South Korean market and completed >10 total knee arthroplasty (TKA) cases using the TSolution One robotic surgical system.
TSolution One received clearance from Korea's Ministry of Food and Drug Safety in 1Q17 for Total Hip Arthroplasty (THA) and TKA. The THA application is FDA 510(k)-cleared and CE Mark-approved,...
THINK Surgical entered the South Korean market and completed >10 total knee arthroplasty (TKA) cases using the TSolution One robotic surgical system.
TSolution One received clearance from Korea’s Ministry of Food and Drug Safety in 1Q17 for Total Hip Arthroplasty (THA) and TKA. The THA application is FDA 510(k)-cleared and CE Mark-approved, and plans are in place to obtain these regulatory clearances for total knee. Worldwide, TSolutions’s technology has been used in thousands of hip and knee cases.
The open-platform system uses CT scans to support 3D pre-surgical planning and offers an alternative to manual template planning, broaching, reaming and impacting techniques for preparation of the femoral canal and acetabular cup. It comprises the TPLAN® Preoperative Planning Workstation and TCAT® robotic system: an electromechanical arm, control electronics and computer, display monitor, cutters, drapes, irrigation sets, etc.
Source: THINK Surgical, Inc.

Image courtesy of THINK Surgical, Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







