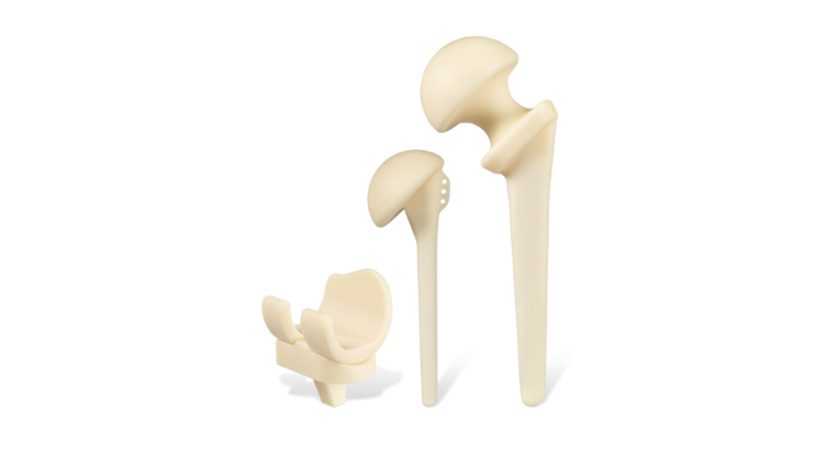
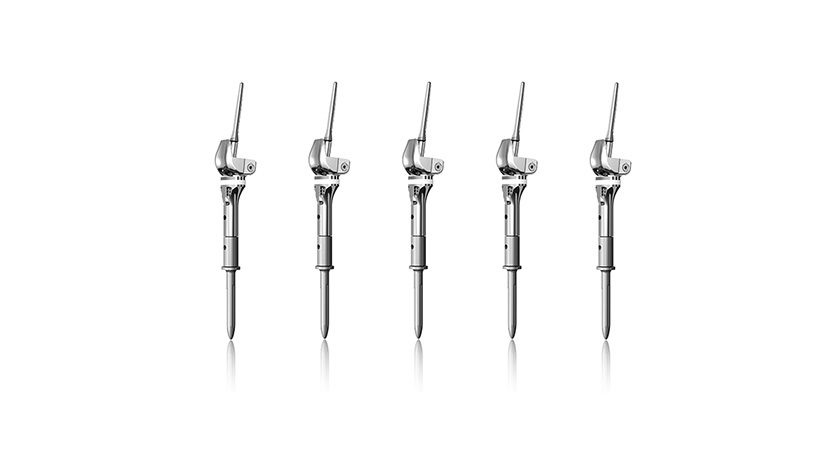
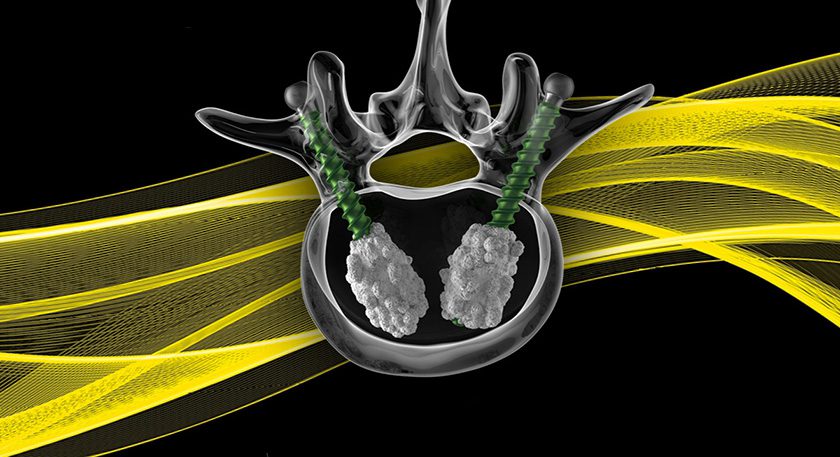
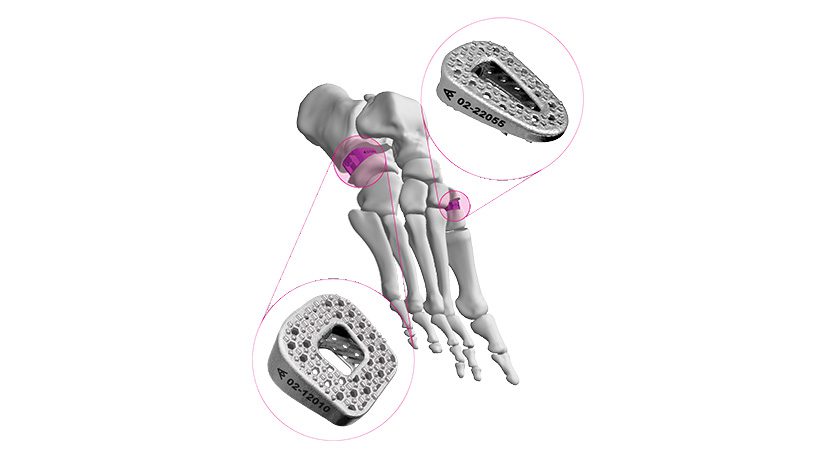
Copy to clipboard
Copy to clipboard 
Three-month safety data from a Phase I clinical trial in China for Cellular Biomedicine’s AlloJoin™ off-the-shelf allogeneic stem cell therapy in the treatment of knee osteoarthritis demonstrated a safety and tolerability profile of AlloJoin in the three doses tested, with adverse events similar to that of earlier autologous trials. The trial is slated for completion in 3Q17.
Summary of trial data:
- No Serious Adverse Events observed for any patients
- Most common Adverse Events are knee pain and swelling after injection, with severity similar to earlier autologous trials (mild to moderate)
Source: Cellular Biomedicine
Three-month safety data from a Phase I clinical trial in China for Cellular Biomedicine's AlloJoin™ off-the-shelf allogeneic stem cell therapy in the treatment of knee osteoarthritis demonstrated a safety and tolerability profile of AlloJoin in the three doses tested, with adverse events similar to that of earlier autologous trials. The trial is...
Three-month safety data from a Phase I clinical trial in China for Cellular Biomedicine’s AlloJoin™ off-the-shelf allogeneic stem cell therapy in the treatment of knee osteoarthritis demonstrated a safety and tolerability profile of AlloJoin in the three doses tested, with adverse events similar to that of earlier autologous trials. The trial is slated for completion in 3Q17.
Summary of trial data:
- No Serious Adverse Events observed for any patients
- Most common Adverse Events are knee pain and swelling after injection, with severity similar to earlier autologous trials (mild to moderate)
Source: Cellular Biomedicine

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.