
 Copy to clipboard
Copy to clipboard 
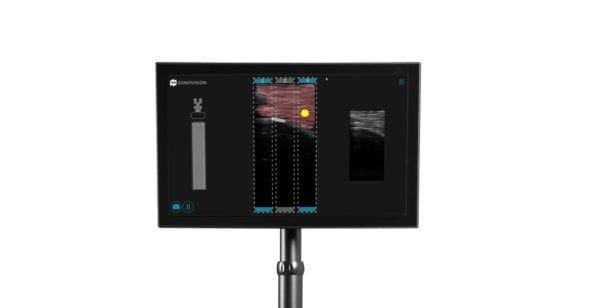
Tissue Differentiation Intelligence (TDi) received FDA 510(k) clearance for the SonoVision™ ultrasound platform, reportedly the first technology of its kind specifically designed for intraoperative access to the spine.
By applying layers of image-processing algorithms to ultrasound images collected intraoperatively, SonoVision visually differentiates nerve, muscle, bone and vessels in real time. The new imaging modality for spine surgery is designed to overcome impracticalities of conventional ultrasound.
TDi has multiple systems deployed for clinical study data collection under Institutional Review Board and for Alpha evaluations, focusing primarily on the clinical refinement of the lateral surgery access product. Expanded applications, including posterior access to the spine, 3D imaging and image-guided navigation integration are in earlier stages of development.
Issued intellectual property protects ultrasonically scanning tissue, interpreting images through signal and image processing, and algorithms used while differentiating tissue based on image characteristics and spectral signatures.
“This is a monumental achievement for TDi which validates the rigor of the research and development process that has occurred over the past 7 years to make this technology possible. With this clearance, TDi ushers in a new era of innovations related to soft tissue imaging in spine surgery, and really, the beginning of a much broader trend of artificial intelligence and machine learning being applied to satisfy challenging clinical requirements in spine surgery,” said Alex Lukianov, TDi Chairman and CEO.
Tissue Differentiation Intelligence (TDi) received FDA 510(k) clearance for the SonoVision™ ultrasound platform, reportedly the first technology of its kind specifically designed for intraoperative access to the spine.
By applying layers of image-processing algorithms to ultrasound images collected intraoperatively, SonoVision visually...
Tissue Differentiation Intelligence (TDi) received FDA 510(k) clearance for the SonoVision™ ultrasound platform, reportedly the first technology of its kind specifically designed for intraoperative access to the spine.
By applying layers of image-processing algorithms to ultrasound images collected intraoperatively, SonoVision visually differentiates nerve, muscle, bone and vessels in real time. The new imaging modality for spine surgery is designed to overcome impracticalities of conventional ultrasound.
TDi has multiple systems deployed for clinical study data collection under Institutional Review Board and for Alpha evaluations, focusing primarily on the clinical refinement of the lateral surgery access product. Expanded applications, including posterior access to the spine, 3D imaging and image-guided navigation integration are in earlier stages of development.
Issued intellectual property protects ultrasonically scanning tissue, interpreting images through signal and image processing, and algorithms used while differentiating tissue based on image characteristics and spectral signatures.
“This is a monumental achievement for TDi which validates the rigor of the research and development process that has occurred over the past 7 years to make this technology possible. With this clearance, TDi ushers in a new era of innovations related to soft tissue imaging in spine surgery, and really, the beginning of a much broader trend of artificial intelligence and machine learning being applied to satisfy challenging clinical requirements in spine surgery,” said Alex Lukianov, TDi Chairman and CEO.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







