
 Copy to clipboard
Copy to clipboard 
Initial outcomes from Spineology’s SCOUT trial of the OptiMesh® device revealed statistically significant improvements in low back pain and function scores at 6 and 12 months.
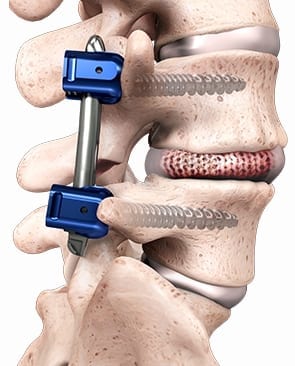
The Spineology Clinical Outcomes Trial, under an FDA-approved Investigational Device Exemption protocol, is a prospective multicenter non-randomized study of instrumented lumbar interbody fusion for the treatment of degenerative disc disease. SCOUT is evaluating the OptiMesh porous graft containment device that deploys within the disc space as it is filled, allowing packed bone graft to conform to the prepared vertebral body endplates. Disc space prep and implant placement are performed through a small cannula.
Of the 102 patients enrolled, interim results derived from 84 treated subjects (60 completing 6-month follow-up and 29 at 12 months). At 12 months all 29 subjects were fused, and 27 of 29 reported “Excellent” or “Good” satisfaction with their surgery. There were no serious device-related adverse events.
The device’s original 2003 FDA 510(k) clearance supported graft containment inside the vertebral body. Data from SCOUT will support the submission of a De Novo application to FDA for expanded indications, allowing use with bone graft and supplemental posterior fixation in lumbar interbody fusion.
Sources: Spineology Inc.; ORTHOWORLD Inc.
Initial outcomes from Spineology's SCOUT trial of the OptiMesh® device revealed statistically significant improvements in low back pain and function scores at 6 and 12 months.
The Spineology Clinical Outcomes Trial, under an FDA-approved Investigational Device Exemption protocol, is a prospective multicenter non-randomized study of...
Initial outcomes from Spineology’s SCOUT trial of the OptiMesh® device revealed statistically significant improvements in low back pain and function scores at 6 and 12 months.
The Spineology Clinical Outcomes Trial, under an FDA-approved Investigational Device Exemption protocol, is a prospective multicenter non-randomized study of instrumented lumbar interbody fusion for the treatment of degenerative disc disease. SCOUT is evaluating the OptiMesh porous graft containment device that deploys within the disc space as it is filled, allowing packed bone graft to conform to the prepared vertebral body endplates. Disc space prep and implant placement are performed through a small cannula.
Of the 102 patients enrolled, interim results derived from 84 treated subjects (60 completing 6-month follow-up and 29 at 12 months). At 12 months all 29 subjects were fused, and 27 of 29 reported “Excellent” or “Good” satisfaction with their surgery. There were no serious device-related adverse events.
The device’s original 2003 FDA 510(k) clearance supported graft containment inside the vertebral body. Data from SCOUT will support the submission of a De Novo application to FDA for expanded indications, allowing use with bone graft and supplemental posterior fixation in lumbar interbody fusion.
Sources: Spineology Inc.; ORTHOWORLD Inc.


You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







