Copy to clipboard
Copy to clipboard 
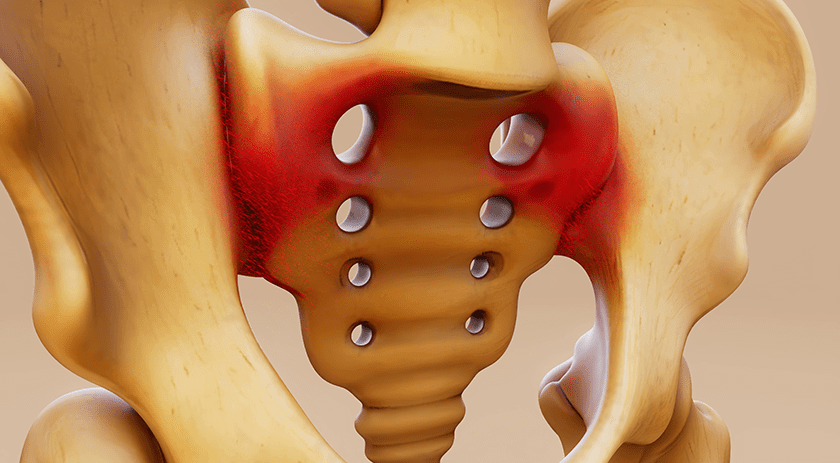
SpineEX gained additional FDA 510(k) clearance for the Sagittae Lateral Lumbar Interbody (LLIF) Fusion system, further advancing the product to full-scale commercialization. First sales are expected within the month.
Sagittae received initial FDA clearance in 4Q18. The expandable device expandable device is available in five sizes designed for up to 8mm of continuous in situ expansion, with up to 30° of continuous in situ lordotic adjustment.
The additional clearance for Sagittae covers new system instrumentation and an updated surgical technique to support streamlined implant sizing and the ability to adjust the anterior and posterior sides independently of one another.
Source: SpineEX
SpineEX gained additional FDA 510(k) clearance for the Sagittae Lateral Lumbar Interbody (LLIF) Fusion system, further advancing the product to full-scale commercialization. First sales are expected within the month.
Sagittae received initial FDA clearance in 4Q18. The expandable device expandable device is available in five sizes designed for...
SpineEX gained additional FDA 510(k) clearance for the Sagittae Lateral Lumbar Interbody (LLIF) Fusion system, further advancing the product to full-scale commercialization. First sales are expected within the month.
Sagittae received initial FDA clearance in 4Q18. The expandable device expandable device is available in five sizes designed for up to 8mm of continuous in situ expansion, with up to 30° of continuous in situ lordotic adjustment.
The additional clearance for Sagittae covers new system instrumentation and an updated surgical technique to support streamlined implant sizing and the ability to adjust the anterior and posterior sides independently of one another.
Source: SpineEX

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.