
 Copy to clipboard
Copy to clipboard 
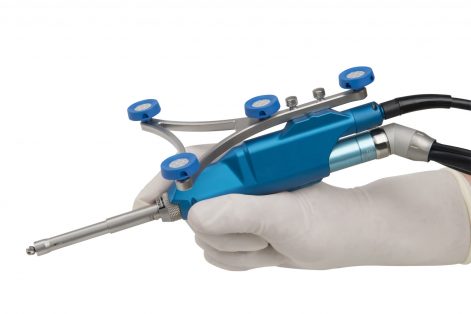
Image courtesy of Smith & Nephew
Smith & Nephew (SNN) announced full launch of its NAVIO handheld robotics-assisted total knee arthroplasty (TKA) application for use with the JOURNEY II, LEGION Primary and GENESIS II systems.
The company received FDA 510(k) clearance for the TKA application in 2Q16, and announced its first surgical use at AAOS 2017, concurrent with launch of Stryker’s Mako total knee application and Zimmer Biomet’s announcement of plans for the Rosa total knee. SNN leadership has stated that NAVIO’s small footprint positions the technology to be a leader in robotic joint reconstruction.
NAVIO’s partial knee replacement application, including unicompartmental and patellofemoral options, has been commercially available since 2014.
Smith & Nephew purchased NAVIO from Blue Belt Technologies in 1Q16 for US $275.0MM. In its 4Q16 earning call, leadership stated that it met NAVIO revenue expectations of growth at +50% over the prior year; Blue Belt’s 2015 revenue was ~$19.0MM.
Sources: Smith & Nephew, ORTHOWORLD Inc.
Smith & Nephew (SNN) announced full launch of its NAVIO handheld robotics-assisted total knee arthroplasty (TKA) application for use with the JOURNEY II, LEGION Primary and GENESIS II systems.
The company received FDA 510(k) clearance for the TKA application in 2Q16, and announced its first surgical use at AAOS 2017, concurrent with launch...

Image courtesy of Smith & Nephew
Smith & Nephew (SNN) announced full launch of its NAVIO handheld robotics-assisted total knee arthroplasty (TKA) application for use with the JOURNEY II, LEGION Primary and GENESIS II systems.
The company received FDA 510(k) clearance for the TKA application in 2Q16, and announced its first surgical use at AAOS 2017, concurrent with launch of Stryker’s Mako total knee application and Zimmer Biomet’s announcement of plans for the Rosa total knee. SNN leadership has stated that NAVIO’s small footprint positions the technology to be a leader in robotic joint reconstruction.
NAVIO’s partial knee replacement application, including unicompartmental and patellofemoral options, has been commercially available since 2014.
Smith & Nephew purchased NAVIO from Blue Belt Technologies in 1Q16 for US $275.0MM. In its 4Q16 earning call, leadership stated that it met NAVIO revenue expectations of growth at +50% over the prior year; Blue Belt’s 2015 revenue was ~$19.0MM.
Sources: Smith & Nephew, ORTHOWORLD Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







