Copy to clipboard
Copy to clipboard 
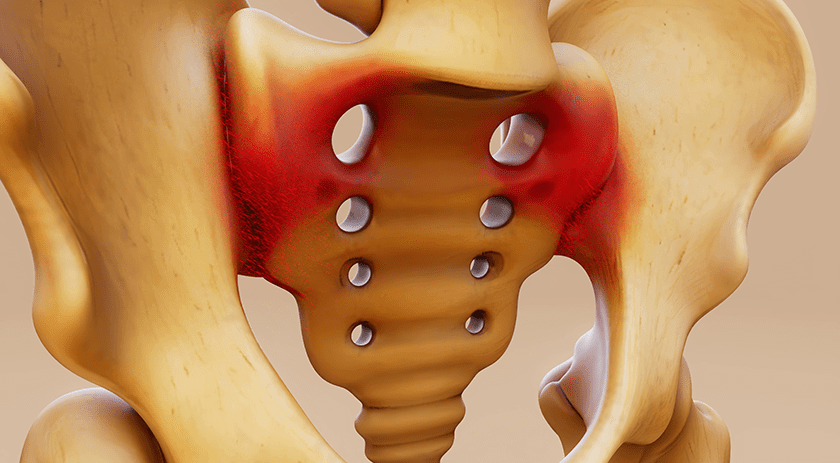
SI-BONE received an additional FDA 510(k) clearance for use of iFuse Bedrock™ technology in fusion of the sacroiliac (SI) joint during long construct procedures. iFuse Bedrock, a spinopelvic fixation solution, is designed to provide greater stabilization of the joint at the base of long spinal constructs.
The clearance supports SI-BONE’s expansion into adjacent markets, such as adult spinal deformity.
iFuse was previously cleared by FDA for SI fusion using a posterior approach to treat SI joint dysfunction in patients also undergoing long fusion procedures involving a posterior approach. With this new broader indication, iFuse Bedrock can be placed across the SI joint as an adjunct to long fusion with the intent of gaining fusion to augment immobilization and stabilization of the SI joint.
Source: SI-BONE
SI-BONE received an additional FDA 510(k) clearance for use of iFuse Bedrock™ technology in fusion of the sacroiliac (SI) joint during long construct procedures. iFuse Bedrock, a spinopelvic fixation solution, is designed to provide greater stabilization of the joint at the base of long spinal constructs.
The clearance supports SI-BONE's...
SI-BONE received an additional FDA 510(k) clearance for use of iFuse Bedrock™ technology in fusion of the sacroiliac (SI) joint during long construct procedures. iFuse Bedrock, a spinopelvic fixation solution, is designed to provide greater stabilization of the joint at the base of long spinal constructs.
The clearance supports SI-BONE’s expansion into adjacent markets, such as adult spinal deformity.
iFuse was previously cleared by FDA for SI fusion using a posterior approach to treat SI joint dysfunction in patients also undergoing long fusion procedures involving a posterior approach. With this new broader indication, iFuse Bedrock can be placed across the SI joint as an adjunct to long fusion with the intent of gaining fusion to augment immobilization and stabilization of the SI joint.
Source: SI-BONE

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.