Copy to clipboard
Copy to clipboard 
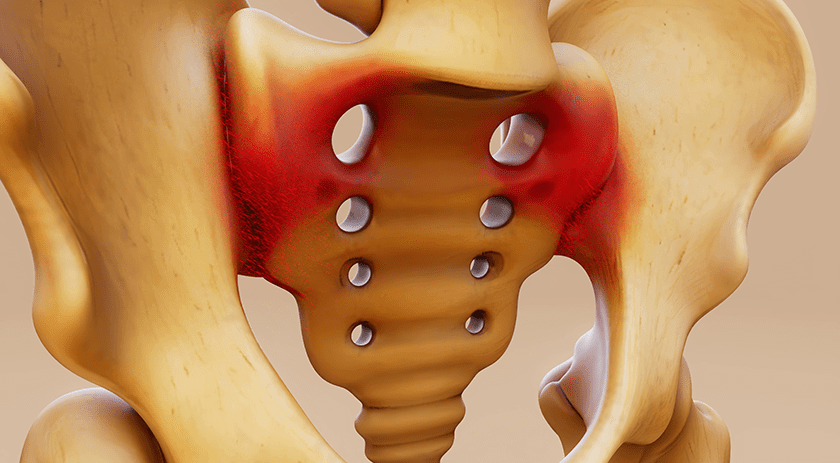
RTI Surgical enrolled the first patient in a clinical evaluation of the Fortilink® interbody fusion device in the treatment of degenerative disc disease.
The study, named FORTE, is a prospective, postmarket evaluation of the safety and performance of the Fortilink-C, -TS, and -L Interbody Fusion systems made with TETRAfuse® 3D printed polymer material. Its primary objective is to assess radiographic evidence of fusion at three months (Fortilink-C IBF) or six months post-op (Fortilink-TS and Fortilink-L IBF).
The FORTE study will collect and evaluate real world data on adult subjects, exploring pain reduction and procedural and long-term performance. The multicenter study will include 150 patients at up to 20 sites in the U.S. over the next 3.5 years, with post-procedure evaluations post-procedurally at intervals throughout 24 months for study data collection. All subjects are expected to be enrolled in 1Q20.
RTI launched the cervical device in 2017; lumbar versions were released in 2018.
Sources: RTI Surgical, Inc.; ORTHOWORLD Inc.
RTI Surgical enrolled the first patient in a clinical evaluation of the Fortilink® interbody fusion device in the treatment of degenerative disc disease.
The study, named FORTE, is a prospective, postmarket evaluation of the safety and performance of the Fortilink-C, -TS, and -L Interbody Fusion systems made with TETRAfuse® 3D printed polymer...
RTI Surgical enrolled the first patient in a clinical evaluation of the Fortilink® interbody fusion device in the treatment of degenerative disc disease.
The study, named FORTE, is a prospective, postmarket evaluation of the safety and performance of the Fortilink-C, -TS, and -L Interbody Fusion systems made with TETRAfuse® 3D printed polymer material. Its primary objective is to assess radiographic evidence of fusion at three months (Fortilink-C IBF) or six months post-op (Fortilink-TS and Fortilink-L IBF).
The FORTE study will collect and evaluate real world data on adult subjects, exploring pain reduction and procedural and long-term performance. The multicenter study will include 150 patients at up to 20 sites in the U.S. over the next 3.5 years, with post-procedure evaluations post-procedurally at intervals throughout 24 months for study data collection. All subjects are expected to be enrolled in 1Q20.
RTI launched the cervical device in 2017; lumbar versions were released in 2018.
Sources: RTI Surgical, Inc.; ORTHOWORLD Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.