
 Copy to clipboard
Copy to clipboard 
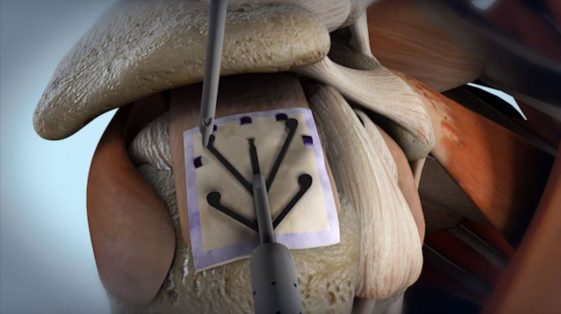
Image courtesy of Rotation Medical
Interim study results indicate that patients receiving Rotation Medical’s collagen-based Bioinductive Implant to treat rotator cuff disease reported statistically significant improvement at six months, in comparison to those undergoing standard treatment at two years.
REBUILD (Rotation MEdical BioindUctive ImpLant Database) is a prospective, non-randomized, multicenter registry collecting patient-reported outcomes (e.g. shoulder function, pain and quality of life) after receiving the Bioinductive Implant. Results from the first 200 patients showed significantly less post-op pain and use of narcotics, less sling time, faster return to function and better overall shoulder rating. The study will follow up to 300 patients at 20 sites.
Previous studies have shown biocompatibility of the implant and its ability to promote new connective tissue with the appearance of tendon, as well as new tissue formation in all study participants with large and massive rotator cuff tears.
Sources: Rotation Medical, Inc.; ORTHOWORLD Inc.
Interim study results indicate that patients receiving Rotation Medical's collagen-based Bioinductive Implant to treat rotator cuff disease reported statistically significant improvement at six months, in comparison to those undergoing standard treatment at two years.
REBUILD (Rotation MEdical BioindUctive ImpLant Database) is a prospective,...

Image courtesy of Rotation Medical
Interim study results indicate that patients receiving Rotation Medical’s collagen-based Bioinductive Implant to treat rotator cuff disease reported statistically significant improvement at six months, in comparison to those undergoing standard treatment at two years.
REBUILD (Rotation MEdical BioindUctive ImpLant Database) is a prospective, non-randomized, multicenter registry collecting patient-reported outcomes (e.g. shoulder function, pain and quality of life) after receiving the Bioinductive Implant. Results from the first 200 patients showed significantly less post-op pain and use of narcotics, less sling time, faster return to function and better overall shoulder rating. The study will follow up to 300 patients at 20 sites.
Previous studies have shown biocompatibility of the implant and its ability to promote new connective tissue with the appearance of tendon, as well as new tissue formation in all study participants with large and massive rotator cuff tears.
Sources: Rotation Medical, Inc.; ORTHOWORLD Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







