
 Copy to clipboard
Copy to clipboard 
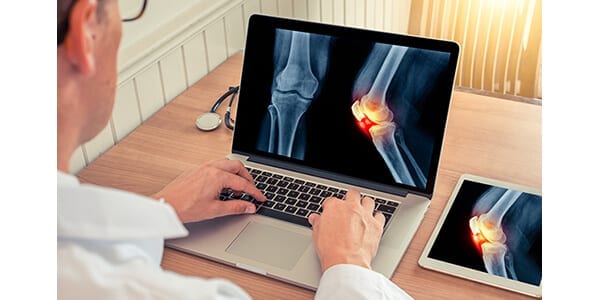
FDA granted 510(k) clearance of Radiobotics’ RBknee™ product, an AI-based analysis tool aimed at improving the efficiency and accuracy of radiographic knee osteoarthritis (OA) diagnosis.
Earlier this year, Radiobotics unveiled its commitment to the U.S. market with its first subsidiary, Radiobotics Inc.
The OA product RBknee works by analyzing digital x-rays of knees and identifying common radiographic findings associated with the diagnosis of OA.
RBknee is intended for use in an orthopedic or a radiological setting, where the x-ray is read in order to determine the diagnosis and treatment of OA. By supporting clinicians with clinically validated AI technology, RBknee has the potential to save time and money as well as improving clinical outcomes through consistent, objective and accurate measurements and analysis.
RBknee can identify presence or absence of osteophytes, subchondral sclerosis, and joint space narrowing using proprietary machine learning algorithms. Furthermore, RBknee can accurately measure the joint space width in both compartments of the knee.
The software is approved on the basis of rigorous testing and clinically validation of the algorithms, showing high sensitivity and specificity on the identification of findings, and a high precision of the measurements of the device when evaluated on an unseen dataset.
“We are proud of this achievement. Getting FDA clearance was always part of our commitment to prove that our product can be used safely at any hospital or clinic across the U.S. Combined with a European MDR certification and CE marking, it reaffirms that we live up to the highest standards and it motivates us to keep working hard to help hospitals and the healthcare industry to move towards a more digitalized future,” said CSO and Co-founder Martin Axelsen.
Source: Radiobiotics
FDA granted 510(k) clearance of Radiobotics’ RBknee™ product, an AI-based analysis tool aimed at improving the efficiency and accuracy of radiographic knee osteoarthritis (OA) diagnosis.
Earlier this year, Radiobotics unveiled its commitment to the U.S. market with its first subsidiary, Radiobotics Inc.
The OA product RBknee works by...
FDA granted 510(k) clearance of Radiobotics’ RBknee™ product, an AI-based analysis tool aimed at improving the efficiency and accuracy of radiographic knee osteoarthritis (OA) diagnosis.
Earlier this year, Radiobotics unveiled its commitment to the U.S. market with its first subsidiary, Radiobotics Inc.
The OA product RBknee works by analyzing digital x-rays of knees and identifying common radiographic findings associated with the diagnosis of OA.
RBknee is intended for use in an orthopedic or a radiological setting, where the x-ray is read in order to determine the diagnosis and treatment of OA. By supporting clinicians with clinically validated AI technology, RBknee has the potential to save time and money as well as improving clinical outcomes through consistent, objective and accurate measurements and analysis.
RBknee can identify presence or absence of osteophytes, subchondral sclerosis, and joint space narrowing using proprietary machine learning algorithms. Furthermore, RBknee can accurately measure the joint space width in both compartments of the knee.
The software is approved on the basis of rigorous testing and clinically validation of the algorithms, showing high sensitivity and specificity on the identification of findings, and a high precision of the measurements of the device when evaluated on an unseen dataset.
“We are proud of this achievement. Getting FDA clearance was always part of our commitment to prove that our product can be used safely at any hospital or clinic across the U.S. Combined with a European MDR certification and CE marking, it reaffirms that we live up to the highest standards and it motivates us to keep working hard to help hospitals and the healthcare industry to move towards a more digitalized future,” said CSO and Co-founder Martin Axelsen.
Source: Radiobiotics

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







