
 Copy to clipboard
Copy to clipboard 
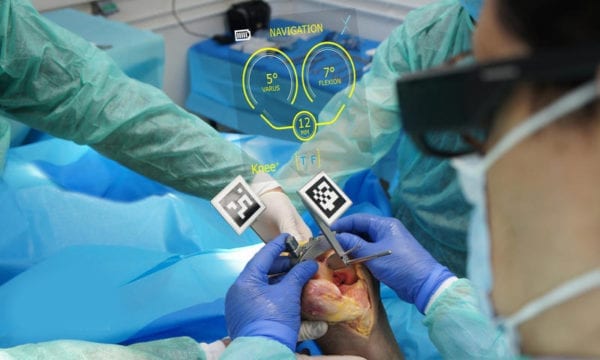
Pixee Medical received CE Mark approval for Knee+, the first orthopedic navigation system to employ augmented reality (AR) for total knee replacement.
The company is pursuing FDA 510(k) clearance for the system, which it seeks to secure within the year.
Knee+ patented technology combines proprietary computer vision and deep learning algorithms, and can be used with off-the-shelf AR glasses to track instruments and implants during a procedure. The navigation software installed in smart glasses is combined with reduced-size MIS instrumentation equipped with markers that can be sterilized in an autoclave. Sizing and ligament balance functions can be quickly integrated into the product.
As an alternative to bulky and expensive robotic systems, Knee+ is simple to use, cost-effective and does not require pre-operative DICOM or disposables.
Pixee Medical has raised over €2.5 million (~USD $2.7 million) and won a grant of €1.2 million (~$1.3 million) to support innovations in computer-assisted orthopedic solutions. Future applications may be introduced for shoulder and hip procedures.
Pixee Medical received CE Mark approval for Knee+, the first orthopedic navigation system to employ augmented reality (AR) for total knee replacement.
The company is pursuing FDA 510(k) clearance for the system, which it seeks to secure within the year.
Knee+ patented technology combines proprietary computer vision and deep learning...
Pixee Medical received CE Mark approval for Knee+, the first orthopedic navigation system to employ augmented reality (AR) for total knee replacement.
The company is pursuing FDA 510(k) clearance for the system, which it seeks to secure within the year.
Knee+ patented technology combines proprietary computer vision and deep learning algorithms, and can be used with off-the-shelf AR glasses to track instruments and implants during a procedure. The navigation software installed in smart glasses is combined with reduced-size MIS instrumentation equipped with markers that can be sterilized in an autoclave. Sizing and ligament balance functions can be quickly integrated into the product.
As an alternative to bulky and expensive robotic systems, Knee+ is simple to use, cost-effective and does not require pre-operative DICOM or disposables.
Pixee Medical has raised over €2.5 million (~USD $2.7 million) and won a grant of €1.2 million (~$1.3 million) to support innovations in computer-assisted orthopedic solutions. Future applications may be introduced for shoulder and hip procedures.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







