
 Copy to clipboard
Copy to clipboard 
PeekMed has received FDA 510(k) clearance to market its previously-CE Marked PeekMed® 3D pre-operative planning software.
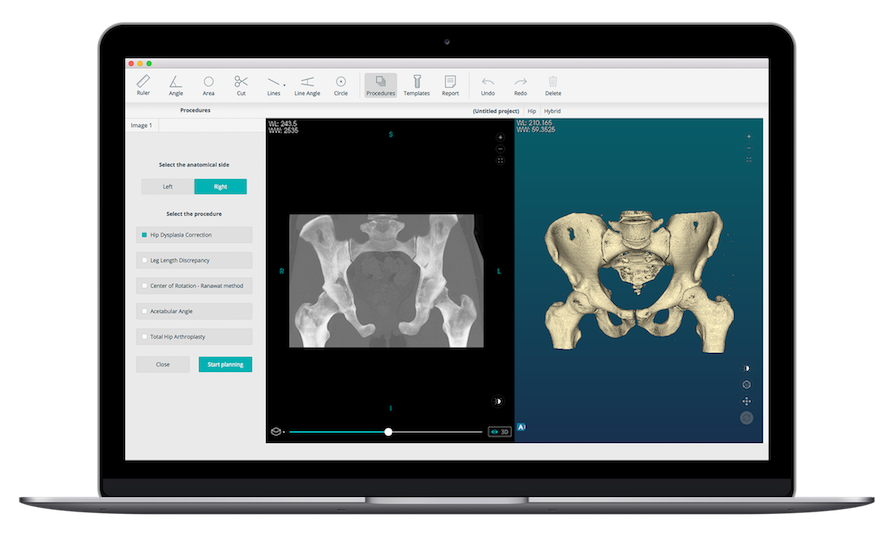 | ||||||
PeekMed uses standard imaging (x-ray, CT, MRI) stored in local PACS servers to allow the surgeon to select from a database of implants to select the option most suitable for the patient.
Other PeekMed features include:
- An intuitive surgical sectioning of a bone in order to correct its deformity. To perform osteotomies, PeekMed includes a wizard and measurement tool specific for each sub-specialty.
- Cloud-based storage for patient studies, data and reports.
- 2D/3D hybrid planning capability.
Current OEM partners with PeekMed include Aesculap, Amplitude Surgical, Arthrex, DePuy Synthes, LimaCorporate, Medtronic, MicroPort, Permedica, Smith & Nephew, Wright Medical and Zimmer Biomet.
Source: PeekMed; image courtesy of PeekMed
PeekMed has received FDA 510(k) clearance to market its previously-CE Marked PeekMed® 3D pre-operative planning software.
PeekMed uses standard imaging (x-ray, CT, MRI) stored in local PACS servers to allow the surgeon to select from a database of implants to select the option most suitable for the...
PeekMed has received FDA 510(k) clearance to market its previously-CE Marked PeekMed® 3D pre-operative planning software.
 | ||||||
PeekMed uses standard imaging (x-ray, CT, MRI) stored in local PACS servers to allow the surgeon to select from a database of implants to select the option most suitable for the patient.
Other PeekMed features include:
- An intuitive surgical sectioning of a bone in order to correct its deformity. To perform osteotomies, PeekMed includes a wizard and measurement tool specific for each sub-specialty.
- Cloud-based storage for patient studies, data and reports.
- 2D/3D hybrid planning capability.
Current OEM partners with PeekMed include Aesculap, Amplitude Surgical, Arthrex, DePuy Synthes, LimaCorporate, Medtronic, MicroPort, Permedica, Smith & Nephew, Wright Medical and Zimmer Biomet.
Source: PeekMed; image courtesy of PeekMed

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







