
 Copy to clipboard
Copy to clipboard 
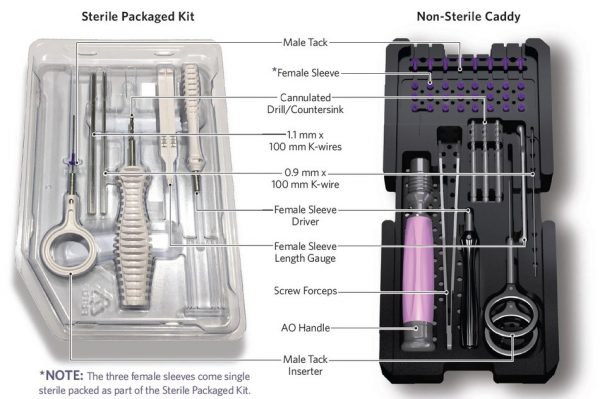
TenoTac™ Sterile and Non-sterile Configurations

Hammertube Caddy Non-sterile and Sterile Configurations – Paragon 28
Paragon 28 launched sterile kit configurations for the TenoTac® Soft Tissue Fixation System for hammertoe and plantar plate repair and the HammerTube™ Hammertoe System.
TenoTac launched in 1Q19 as the first dedicated system to address tendon transfers of the forefoot. As opposed to traditional flexor tendon transfer methods, TenoTac does not release tissue attachments distally but rather by balancing plantar and dorsal tension. Cannulated implants and instrumentation support a reproducible technique with minimal steps.
A titanium threaded implant and simple insertion method are employed to gain fixation of soft tissue to bone. The phalanx is prepped with the provided instrumentation and the plantar tack is inserted into the bone tunnel, capturing plantar tendons. Once inserted, temporary fixation and correction are achieved by holding manual tension over the tendon and bone. While maintaining this tension, the dorsal sleeve of the implant is mated to the plantar tack by threading the two components together, capturing the tendon plantarly and completing the correction.
HammerTube, launched in 1Q18, is a single-piece titanium-sprayed PEEK implant for use in proximal interphalangeal joint fusion. The system provides four implants in straight and angled options. The tapered design facilitates insertion and a press fit.
The sterile kit configurations eliminate the need for pre-operative sterilization and simplify the back table for surgical techs and surgeons. The kits can be available in the O.R. at a moment’s notice.
Paragon 28 launched sterile kit configurations for the TenoTac® Soft Tissue Fixation System for hammertoe and plantar plate repair and the HammerTube™ Hammertoe System.
TenoTac launched in 1Q19 as the first dedicated system to address tendon transfers of the forefoot. As opposed to traditional flexor tendon transfer methods, TenoTac does...

TenoTac™ Sterile and Non-sterile Configurations

Hammertube Caddy Non-sterile and Sterile Configurations – Paragon 28
Paragon 28 launched sterile kit configurations for the TenoTac® Soft Tissue Fixation System for hammertoe and plantar plate repair and the HammerTube™ Hammertoe System.
TenoTac launched in 1Q19 as the first dedicated system to address tendon transfers of the forefoot. As opposed to traditional flexor tendon transfer methods, TenoTac does not release tissue attachments distally but rather by balancing plantar and dorsal tension. Cannulated implants and instrumentation support a reproducible technique with minimal steps.
A titanium threaded implant and simple insertion method are employed to gain fixation of soft tissue to bone. The phalanx is prepped with the provided instrumentation and the plantar tack is inserted into the bone tunnel, capturing plantar tendons. Once inserted, temporary fixation and correction are achieved by holding manual tension over the tendon and bone. While maintaining this tension, the dorsal sleeve of the implant is mated to the plantar tack by threading the two components together, capturing the tendon plantarly and completing the correction.
HammerTube, launched in 1Q18, is a single-piece titanium-sprayed PEEK implant for use in proximal interphalangeal joint fusion. The system provides four implants in straight and angled options. The tapered design facilitates insertion and a press fit.
The sterile kit configurations eliminate the need for pre-operative sterilization and simplify the back table for surgical techs and surgeons. The kits can be available in the O.R. at a moment’s notice.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







