Copy to clipboard
Copy to clipboard 
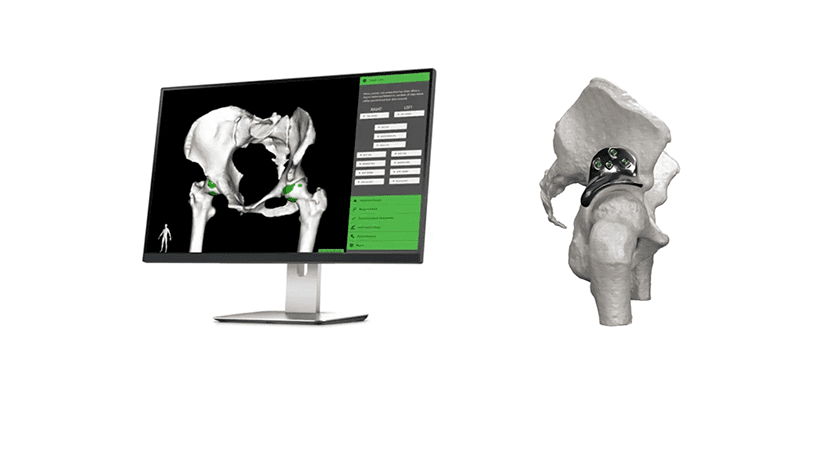
Paragon 28 completed a Series B financing. Funds will support ongoing initiatives to expand global expansion and develop and launch new products. This includes the APEX 3D™ Total Ankle Replacement (pictured above) that gained FDA 510(k) clearance in July 2020.
Founded in 2010, the company focuses exclusively upon specialization and innovation of plates, screws, etc. to treat fractures, fusion, flatfoot, hammertoe and more in the 28 bones of the foot. APEX 3D represents their entry into the total joint replacement market.
In 2011, the company initialized its first capital raise of $200,000.
Albert DaCosta, President and CEO of Paragon 28, said, “We are pleased to add this capital to our balance sheet and equally excited about the partnership. The Series B proceeds will enable our team to focus on the best growth opportunities and expand our market position. To close this round on the tenth anniversary of the company is a special moment for our team.”
According to Frank Bono, CTO and CO-Founder of Paragon 28, the company targets annual revenue of $1 billion.
Piper Sandler Merchant Banking and MVM Partners co-led the financing round, including participation from an existing Series A investor.
Paragon 28 completed a Series B financing. Funds will support ongoing initiatives to expand global expansion and develop and launch new products. This includes the APEX 3D™ Total Ankle Replacement (pictured above) that gained FDA 510(k) clearance in July 2020.
Founded in 2010, the company focuses exclusively upon specialization and...
Paragon 28 completed a Series B financing. Funds will support ongoing initiatives to expand global expansion and develop and launch new products. This includes the APEX 3D™ Total Ankle Replacement (pictured above) that gained FDA 510(k) clearance in July 2020.
Founded in 2010, the company focuses exclusively upon specialization and innovation of plates, screws, etc. to treat fractures, fusion, flatfoot, hammertoe and more in the 28 bones of the foot. APEX 3D represents their entry into the total joint replacement market.
In 2011, the company initialized its first capital raise of $200,000.
Albert DaCosta, President and CEO of Paragon 28, said, “We are pleased to add this capital to our balance sheet and equally excited about the partnership. The Series B proceeds will enable our team to focus on the best growth opportunities and expand our market position. To close this round on the tenth anniversary of the company is a special moment for our team.”
According to Frank Bono, CTO and CO-Founder of Paragon 28, the company targets annual revenue of $1 billion.
Piper Sandler Merchant Banking and MVM Partners co-led the financing round, including participation from an existing Series A investor.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.