Copy to clipboard
Copy to clipboard 
Panther Orthopedics closed a Series B financing, led by Medeon Biodesign, and has expanded clinical use of its PUMA™ dynamic fixation system.
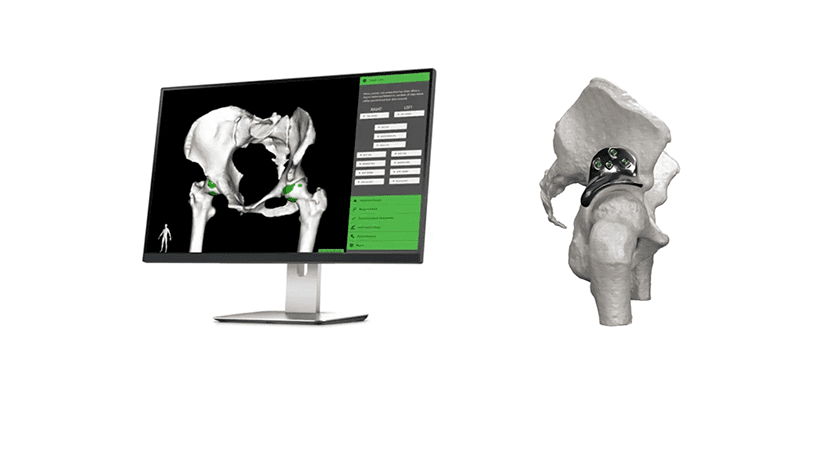
PUMA obtained FDA 510(k) clearance in early 2018 for indications of syndesmosis fixation, hallux valgus reconstruction and tarsometatarsal fixation, and was commercially launched in the U.S. later that year. A spring-like Nitinol body design provides initial and continuous compression without the need to overtighten.
With additional financing backing, Panther Orthopedics can now continue to expand its clinical and commercial operations.
Medeon Biodesign, an incubator group based in Taiwan, develops medical devices for minimally invasive procedures across a range of specialties, including orthopaedics. As the lead Series B investor, Medeon will provide resources such as funding, development expertise, manufacturing capabilities, etc. to accelerate Panther’s development.
Panther Orthopedics closed a Series B financing, led by Medeon Biodesign, and has expanded clinical use of its PUMA™ dynamic fixation system.
PUMA obtained FDA 510(k) clearance in early 2018 for indications of syndesmosis fixation, hallux valgus reconstruction and tarsometatarsal fixation, and was commercially launched in the U.S. later...
Panther Orthopedics closed a Series B financing, led by Medeon Biodesign, and has expanded clinical use of its PUMA™ dynamic fixation system.
PUMA obtained FDA 510(k) clearance in early 2018 for indications of syndesmosis fixation, hallux valgus reconstruction and tarsometatarsal fixation, and was commercially launched in the U.S. later that year. A spring-like Nitinol body design provides initial and continuous compression without the need to overtighten.
With additional financing backing, Panther Orthopedics can now continue to expand its clinical and commercial operations.
Medeon Biodesign, an incubator group based in Taiwan, develops medical devices for minimally invasive procedures across a range of specialties, including orthopaedics. As the lead Series B investor, Medeon will provide resources such as funding, development expertise, manufacturing capabilities, etc. to accelerate Panther’s development.
Source: Panther Orthopedics/Medeon Biodesign, Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.