
 Copy to clipboard
Copy to clipboard 
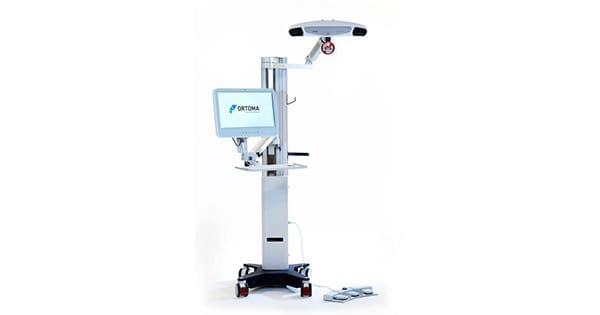
Ortoma obtained approval under the CE Mark for its spine module, OTS Spine Plan, now included in the Ortoma Treatment Solution (OTS) platform for pre-operative surgical planning.
The OTS Spine Plan is built on the OTS platform that is assisted by AI, and is designed to address the challenges of the spine surgery market.
The OTS platform now includes CE-marked products to support hip replacement and spine surgery. The OTS for hip replacement is FDA 510(k) cleared and marketed in the U.S.
“OTS Spine Plan is an important extension of OTS and proves the scalability and flexibility of the AI assisted platform. With the CE-marked product, we are ready to take the next step and offer the product for use clinically,” said Linus Byström, CEO of Ortoma.
Ortoma obtained approval under the CE Mark for its spine module, OTS Spine Plan, now included in the Ortoma Treatment Solution (OTS) platform for pre-operative surgical planning.
The OTS Spine Plan is built on the OTS platform that is assisted by AI, and is designed to address the challenges of the spine surgery market.
The OTS platform...
Ortoma obtained approval under the CE Mark for its spine module, OTS Spine Plan, now included in the Ortoma Treatment Solution (OTS) platform for pre-operative surgical planning.
The OTS Spine Plan is built on the OTS platform that is assisted by AI, and is designed to address the challenges of the spine surgery market.
The OTS platform now includes CE-marked products to support hip replacement and spine surgery. The OTS for hip replacement is FDA 510(k) cleared and marketed in the U.S.
“OTS Spine Plan is an important extension of OTS and proves the scalability and flexibility of the AI assisted platform. With the CE-marked product, we are ready to take the next step and offer the product for use clinically,” said Linus Byström, CEO of Ortoma.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







