
 Copy to clipboard
Copy to clipboard 
OrthoXel received FDA 510(k) clearance to market the Apex Femoral Nailing System. Launch is slated for 2019. This follows the regulatory clearance and first clinical use of the company’s first product, a tibial nail, in 1Q18.
The universal nail features anatomic nail curvature and can be implanted from antegrade or retrograde orientations. Multiple-trajectory locking options include OrthoXel’s exclusive micromotion for controlled axial movement with torsional stability that promotes callus formation. As needed, a locking endcap simultaneously locks all screws.
Sources: OrthoXel; ORTHOWORLD Inc.
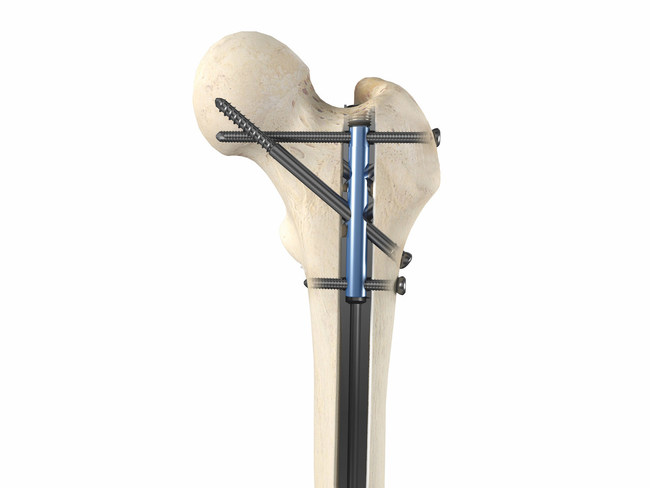
Image courtesy of OrthoXel
OrthoXel received FDA 510(k) clearance to market the Apex Femoral Nailing System. Launch is slated for 2019. This follows the regulatory clearance and first clinical use of the company's first product, a tibial nail, in 1Q18.
The universal nail features anatomic nail curvature and can be implanted from antegrade or retrograde...
OrthoXel received FDA 510(k) clearance to market the Apex Femoral Nailing System. Launch is slated for 2019. This follows the regulatory clearance and first clinical use of the company’s first product, a tibial nail, in 1Q18.
The universal nail features anatomic nail curvature and can be implanted from antegrade or retrograde orientations. Multiple-trajectory locking options include OrthoXel’s exclusive micromotion for controlled axial movement with torsional stability that promotes callus formation. As needed, a locking endcap simultaneously locks all screws.
Sources: OrthoXel; ORTHOWORLD Inc.

Image courtesy of OrthoXel

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







