
 Copy to clipboard
Copy to clipboard 
Orthox raised a further $4.3 million to close its $12.5 million Series A financing round. Proceeds will support additional clinical trials of its lead product, FibroFix™, in Hungary and the U.K.
Recruitment for the pivotal clinical trial will open later this year, with a first cohort of six patients and six-month follow-up. This will demonstrate initial safety. Subsequently, the trial will be expanded to 75 patients with two years of follow-up.
FibroFix technology is a tissue scaffold based on fibroin, a tissue regenerative protein extracted from silk fibers. The implants remove only damaged tissue, employing bone-sparing, minimally invasive surgical techniques to support rapid patient rehabilitation.
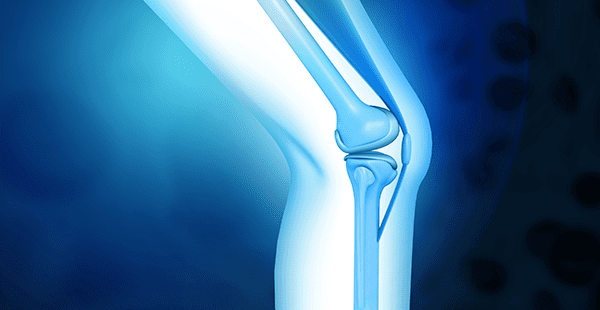
FibroFix treats injuries to the articular cartilage in the knee, which are often sports-related. It protects the surface of these bones and allows the joint to operate smoothly. The procedure offers an alternative to debridement and microfracture.
CEO Nick Skaer said, “FibroFix is unique in emulating the functional properties of cartilage while also facilitating rapid tissue regeneration in patients suffering the debilitating effects of serious knee cartilage injuries. With our approach there is significant bone sparing and strong tissue integration of the implant, which will result in much faster patient recovery from surgery.”
Source: Orthox
Orthox raised a further $4.3 million to close its $12.5 million Series A financing round. Proceeds will support additional clinical trials of its lead product, FibroFix™, in Hungary and the U.K.
Recruitment for the pivotal clinical trial will open later this year, with a first cohort of six patients and six-month follow-up. This will demonstrate...
Orthox raised a further $4.3 million to close its $12.5 million Series A financing round. Proceeds will support additional clinical trials of its lead product, FibroFix™, in Hungary and the U.K.
Recruitment for the pivotal clinical trial will open later this year, with a first cohort of six patients and six-month follow-up. This will demonstrate initial safety. Subsequently, the trial will be expanded to 75 patients with two years of follow-up.
FibroFix technology is a tissue scaffold based on fibroin, a tissue regenerative protein extracted from silk fibers. The implants remove only damaged tissue, employing bone-sparing, minimally invasive surgical techniques to support rapid patient rehabilitation.
FibroFix treats injuries to the articular cartilage in the knee, which are often sports-related. It protects the surface of these bones and allows the joint to operate smoothly. The procedure offers an alternative to debridement and microfracture.
CEO Nick Skaer said, “FibroFix is unique in emulating the functional properties of cartilage while also facilitating rapid tissue regeneration in patients suffering the debilitating effects of serious knee cartilage injuries. With our approach there is significant bone sparing and strong tissue integration of the implant, which will result in much faster patient recovery from surgery.”
Source: Orthox

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.