
 Copy to clipboard
Copy to clipboard 
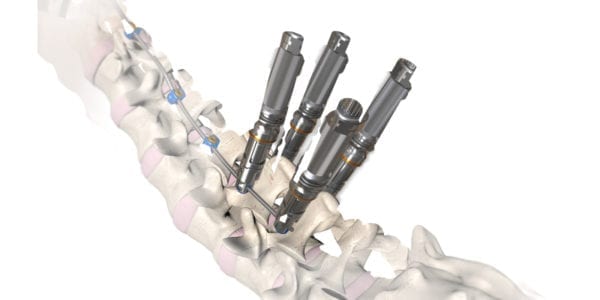
OrthoPediatrics was granted FDA 510(k) clearance to expand the indications for its RESPONSE™ Scoliosis System to include neuromuscular implants.
RESPONSE™ NeuroMuscular is based on the company’s RESPONSE™ Spine System, and will feature implants and instruments with specific attributes to simplify insertion and options to address extreme hyperlordosis.
Executive Vice President David Bailey said, “While we cannot provide our expected launch timeline given the current environment, we remain undeterred on our cause of improving the lives of children suffering from scoliosis. I’m pleased by the resolve of all our associates who continue to advance our business while working remotely, so that we can emerge from the COVID-19 crisis even stronger together.”
OrthoPediatrics was granted FDA 510(k) clearance to expand the indications for its RESPONSE™ Scoliosis System to include neuromuscular implants.
RESPONSE™ NeuroMuscular is based on the company’s RESPONSE™ Spine System, and will feature implants and instruments with specific attributes to simplify insertion and options to address extreme...
OrthoPediatrics was granted FDA 510(k) clearance to expand the indications for its RESPONSE™ Scoliosis System to include neuromuscular implants.
RESPONSE™ NeuroMuscular is based on the company’s RESPONSE™ Spine System, and will feature implants and instruments with specific attributes to simplify insertion and options to address extreme hyperlordosis.
Executive Vice President David Bailey said, “While we cannot provide our expected launch timeline given the current environment, we remain undeterred on our cause of improving the lives of children suffering from scoliosis. I’m pleased by the resolve of all our associates who continue to advance our business while working remotely, so that we can emerge from the COVID-19 crisis even stronger together.”

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







