ORTHOFLASH®
The range of spinal products and technologies provides a modular workflow for posterior stabilization and deformity correction.
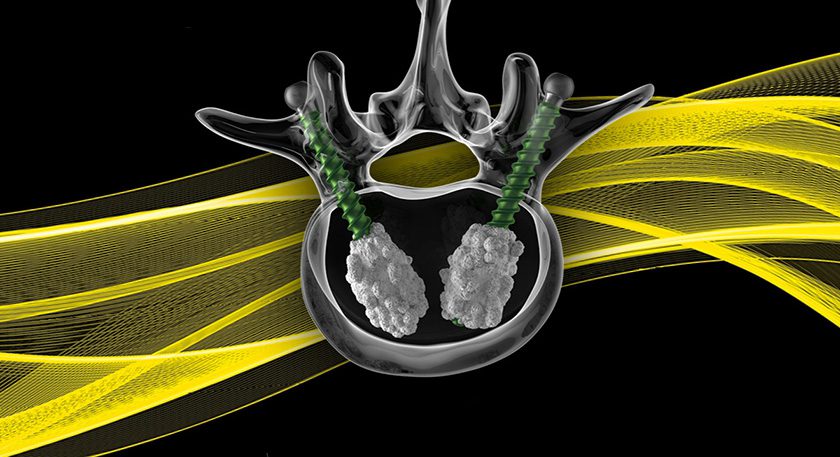
ARKEO is designed to optimize fixation and graft integration through anatomical fit, open architecture, and controlled stability.
The Phase 3 trial is evaluating the single-injection biologic candidate for improvements in pain and function.
October 14, 2025
,
Julie A. Vetalice
October 10, 2025
,
Patrick McGuire
The range of spinal products and technologies provides a modular workflow for posterior stabilization and deformity correction.
October 20, 2025
,
ARKEO is designed to optimize fixation and graft integration through anatomical fit, open architecture, and controlled stability.
October 17, 2025
,
The Phase 3 trial is evaluating the single-injection biologic candidate for improvements in pain and function.
October 16, 2025
,
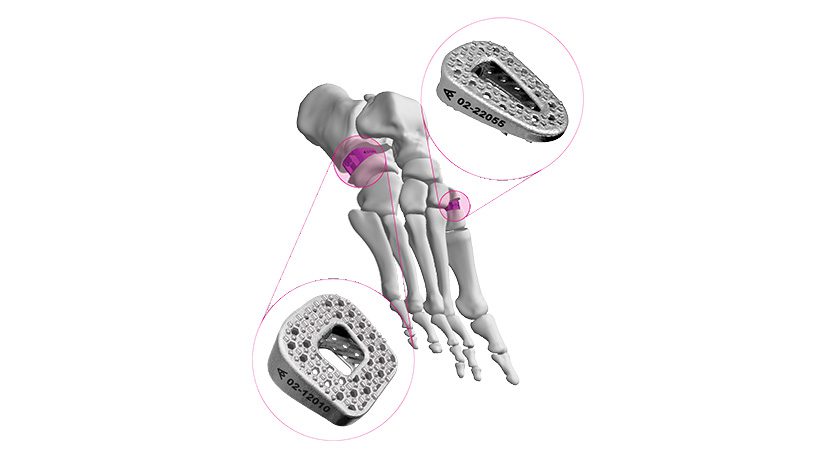
ANTHEM Elbow is designed to offer surgeons anatomically-contoured plates to treat a variety of fractures, regardless of severity or complexity,
October 15, 2025
,
The prodisc C Vivo and prodisc C SK are now indicated for reconstruction of a vertebral disc from C3-C7 following discectomy at one level or two contiguous levels.
October 14, 2025
,
The total shoulder replacement system is designed for tissue-sparing surgery, providing same day post-op mobility.
October 13, 2025
,
SyncAR integrates with Medtronic StealthStation navigation and incorporates AI-driven vertebra segmentation with various elements to keep pre-op models aligned with intra-op CT scans.
October 10, 2025
,
The code recognizes the clinical significance and growing adoption of CARTIHEAL, a single-stage, off-the-shelf solution to treat cartilage and osteochondral defects in the knee.
October 9, 2025
,
The real-world study confirms long-term safety and efficacy of minimally invasive posterior SI joint fusion using novel cortical allograft.
October 8, 2025
,
The company announced the full commercial launch of the Gorilla Pilon Fusion Plating System and the Phantom TTC Trauma Nail.
October 8, 2025
,
ALAYA is a CE-certified surgical robotic assistant for spine that features patented Kinematic Navigation Technology.
October 8, 2025
,
Study participants received the company’s next generation CEM-Plate and CEM-Cage used in anterior cervical discectomy and fusion procedures.
October 7, 2025
,
TigerShark Lateral uses the proprietary BioBond porous trabecular structure to create the new lateral interbody system.
October 6, 2025
,
Introduction of the pre-op planning and modeling system completes the CORIOGRAPH Services portfolio, which includes solutions for knee and hip arthroplasty.
October 2, 2025
,
Bioretec’s RemeOs screw is the first and only osteopromotive absorbable metal implant for orthopedic use in the U.S.