Copy to clipboard
Copy to clipboard 
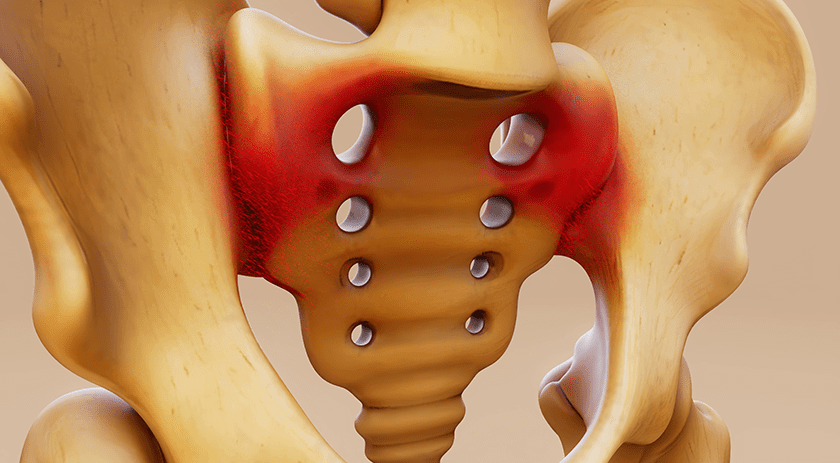
NuVasive’s proprietary Cohere® Porous polyetheretherketone (PEEK) implant demonstrated clear benefits in promoting improved early outcomes in Anterior Cervical Discectomy and Fusion (ACDF) when compared to structural allograft and smooth PEEK interbody devices.
In the study, 167 patients with degenerative cervical disc disease underwent ACDF using Cohere Porous PEEK, structural allograft or smooth PEEK in the interbody space. Those receiving Cohere exhibited significant clinical improvements over pre-op disability and pain scores.
Specifically, these improvements were observed at six weeks post-op, while patients treated with smooth PEEK did not show significant improvements until three months post-op. While structural allograft patients began to improve over baseline by six weeks, Cohere patients showed statistically significant improvements over baseline by two weeks and significantly greater improvements than allograft and smooth PEEK patients by six weeks post-op. This early improvement was maintained at all reported post-op time points out to one year.
Source: NuVasive, Inc.
NuVasive's proprietary Cohere® Porous polyetheretherketone (PEEK) implant demonstrated clear benefits in promoting improved early outcomes in Anterior Cervical Discectomy and Fusion (ACDF) when compared to structural allograft and smooth PEEK interbody devices.
In the study, 167 patients with degenerative cervical disc disease underwent ACDF...
NuVasive’s proprietary Cohere® Porous polyetheretherketone (PEEK) implant demonstrated clear benefits in promoting improved early outcomes in Anterior Cervical Discectomy and Fusion (ACDF) when compared to structural allograft and smooth PEEK interbody devices.
In the study, 167 patients with degenerative cervical disc disease underwent ACDF using Cohere Porous PEEK, structural allograft or smooth PEEK in the interbody space. Those receiving Cohere exhibited significant clinical improvements over pre-op disability and pain scores.
Specifically, these improvements were observed at six weeks post-op, while patients treated with smooth PEEK did not show significant improvements until three months post-op. While structural allograft patients began to improve over baseline by six weeks, Cohere patients showed statistically significant improvements over baseline by two weeks and significantly greater improvements than allograft and smooth PEEK patients by six weeks post-op. This early improvement was maintained at all reported post-op time points out to one year.
Source: NuVasive, Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.