
 Copy to clipboard
Copy to clipboard 
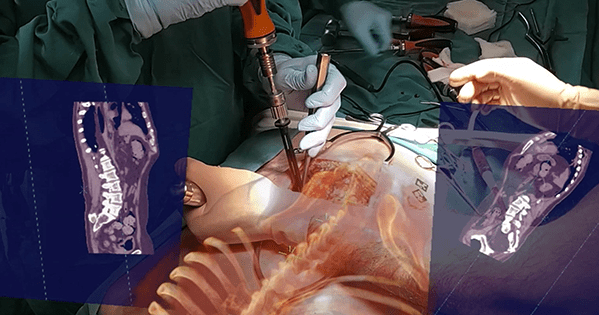
VisAR, an augmented reality surgical navigation system from Novarad, received FDA 510(k) clearance for precision-guided intraoperative spine surgery. VisAR transforms a patient’s imaging data into a 3-dimensional hologram which is visible through an optical visor, and superimposed onto the patient with submillimeter accuracy. This allows the surgeon to focus directly on the surgical objective without looking away at a separate monitor.
VisAR is an end-to-end offering with pre-surgical planning, virtual annotations, segmentation and bi-directional image connectivity. It features integrated 2D and 3D immersive navigation views with continuous hologram-to-patient registration. VisAR technology utilizes image visible CT fiducial markers for automatic registration. The operating room setup time is less than 2 minutes. Surgical accuracy is sub 2 mm for pedicle screw placement in both open and minimally invasive surgical procedures.
Novarad has partnered with Microsoft to utilize off-the-shelf AR headset technology which allows for lowered cost and the ability to leverage expected hardware advancements. The untethered wirelessly connected Microsoft HoloLens 2 visor worn by the physician results in the smallest OR footprint of any system on the market. No other navigation equipment is required. VisAR is built on the Novarad imaging technology solution stack that provides interoperability, HIPAA compliance, image management and deep security.
VisAR is currently available in the U.S., with usage being anticipated in other countries in the coming months.
Source: Novarad
VisAR, an augmented reality surgical navigation system from Novarad, received FDA 510(k) clearance for precision-guided intraoperative spine surgery. VisAR transforms a patient's imaging data into a 3-dimensional hologram which is visible through an optical visor, and superimposed onto the patient with submillimeter accuracy. This allows the...
VisAR, an augmented reality surgical navigation system from Novarad, received FDA 510(k) clearance for precision-guided intraoperative spine surgery. VisAR transforms a patient’s imaging data into a 3-dimensional hologram which is visible through an optical visor, and superimposed onto the patient with submillimeter accuracy. This allows the surgeon to focus directly on the surgical objective without looking away at a separate monitor.
VisAR is an end-to-end offering with pre-surgical planning, virtual annotations, segmentation and bi-directional image connectivity. It features integrated 2D and 3D immersive navigation views with continuous hologram-to-patient registration. VisAR technology utilizes image visible CT fiducial markers for automatic registration. The operating room setup time is less than 2 minutes. Surgical accuracy is sub 2 mm for pedicle screw placement in both open and minimally invasive surgical procedures.
Novarad has partnered with Microsoft to utilize off-the-shelf AR headset technology which allows for lowered cost and the ability to leverage expected hardware advancements. The untethered wirelessly connected Microsoft HoloLens 2 visor worn by the physician results in the smallest OR footprint of any system on the market. No other navigation equipment is required. VisAR is built on the Novarad imaging technology solution stack that provides interoperability, HIPAA compliance, image management and deep security.
VisAR is currently available in the U.S., with usage being anticipated in other countries in the coming months.
Source: Novarad

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







