
 Copy to clipboard
Copy to clipboard 
Naviswiss received FDA 510(k) clearance to market Naviplan. Naviswiss is releasing the CT-based Navigation and Naviplan to orthopedic care centers in the U.S. over the course of 4Q21.
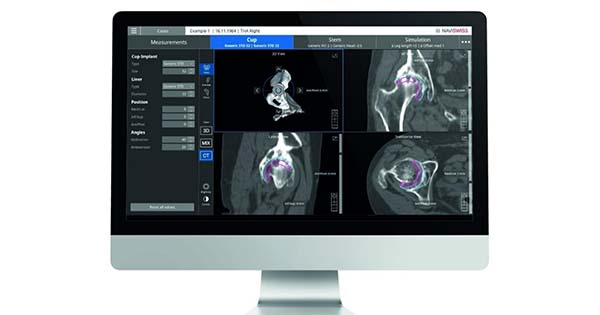
Naviplan is a digital pre-operative planning application enabling orthopedic surgeons to perform navigated CT-based total hip replacement surgery. This has the potential to improve accuracy and predictability and provides a seamless documentation of the outcome.
The Naviplan hip application is CT Based and assists the surgeon in the optimal positioning of the joint implants, automatic 3D segmentation and advanced image processing. Naviplan outputs the pre-operative plan into the Naviswiss navigation platform for accurate surgical execution.
“Naviplan and CT-based Navigation are an important addition to the Naviswiss portfolio and completes our offering for navigated hip replacement”, said Jan Stifter, Naviswiss CEO. “We now have two patient specific options where the surgeon determines the best application for the procedure. CT-based Navigation may be needed in difficult deformity cases while kinematic registration may be preferred in more traditional surgeries. The surgeon can rely on highly accurate guidance in placing the acetabular components.”
Source: Naviswiss
Naviswiss received FDA 510(k) clearance to market Naviplan. Naviswiss is releasing the CT-based Navigation and Naviplan to orthopedic care centers in the U.S. over the course of 4Q21.
Naviplan is a digital pre-operative planning application enabling orthopedic surgeons to perform navigated CT-based total hip replacement surgery. This has the...
Naviswiss received FDA 510(k) clearance to market Naviplan. Naviswiss is releasing the CT-based Navigation and Naviplan to orthopedic care centers in the U.S. over the course of 4Q21.
Naviplan is a digital pre-operative planning application enabling orthopedic surgeons to perform navigated CT-based total hip replacement surgery. This has the potential to improve accuracy and predictability and provides a seamless documentation of the outcome.
The Naviplan hip application is CT Based and assists the surgeon in the optimal positioning of the joint implants, automatic 3D segmentation and advanced image processing. Naviplan outputs the pre-operative plan into the Naviswiss navigation platform for accurate surgical execution.
“Naviplan and CT-based Navigation are an important addition to the Naviswiss portfolio and completes our offering for navigated hip replacement”, said Jan Stifter, Naviswiss CEO. “We now have two patient specific options where the surgeon determines the best application for the procedure. CT-based Navigation may be needed in difficult deformity cases while kinematic registration may be preferred in more traditional surgeries. The surgeon can rely on highly accurate guidance in placing the acetabular components.”
Source: Naviswiss

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







