Copy to clipboard
Copy to clipboard 
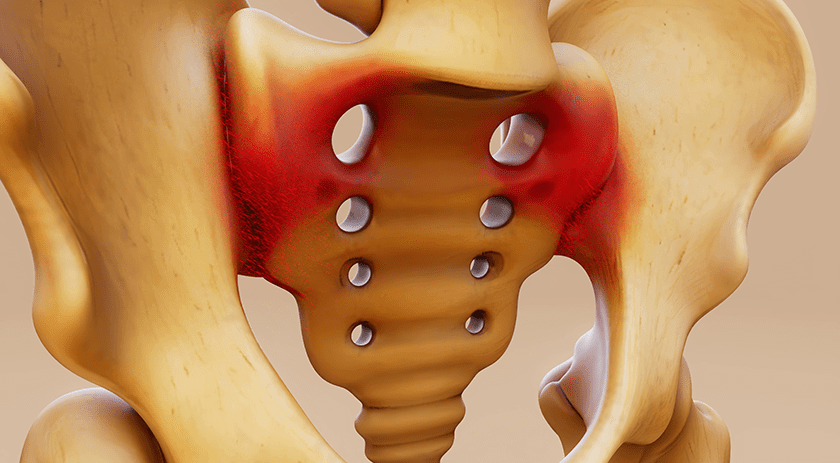
MiRus received FDA 510(k) clearance for Europa™, a pedicle screw made with the company’s proprietary MoRe® molybdenum rhenium superalloy.
This marks the first FDA clearance of a medical device with this material. MiRus developed it to provide strength, ductility and durability surpassing the limitations of titanium, cobalt and iron based alloys. Its use may help address issues such as rod failure in spinal deformity procedures, and could enable the creation of lower profile foot and ankle implants, leading to smaller surgical exposures and lower revision rates.
Source: MiRus LLC
MiRus received FDA 510(k) clearance for Europa™, a pedicle screw made with the company's proprietary MoRe® molybdenum rhenium superalloy.
This marks the first FDA clearance of a medical device with this material. MiRus developed it to provide strength, ductility and durability surpassing the limitations of titanium, cobalt and iron based...
MiRus received FDA 510(k) clearance for Europa™, a pedicle screw made with the company’s proprietary MoRe® molybdenum rhenium superalloy.
This marks the first FDA clearance of a medical device with this material. MiRus developed it to provide strength, ductility and durability surpassing the limitations of titanium, cobalt and iron based alloys. Its use may help address issues such as rod failure in spinal deformity procedures, and could enable the creation of lower profile foot and ankle implants, leading to smaller surgical exposures and lower revision rates.
Source: MiRus LLC

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.