Copy to clipboard
Copy to clipboard 
 | |||||
MIACH Orthopaedics closed a US $22.5MM round of Series A financing. Funds will support accelerated manufacturing and continued clinical trials for the company’s first implant, Bridge-Enhanced® ACL Repair (BEAR®), designed to stimulate healing of torn anterior cruciate ligaments (ACL).
The round was co-led by Amzak Health Investors LLC and DSM Venturing, with participation from the NFL Players Association and additional investors.
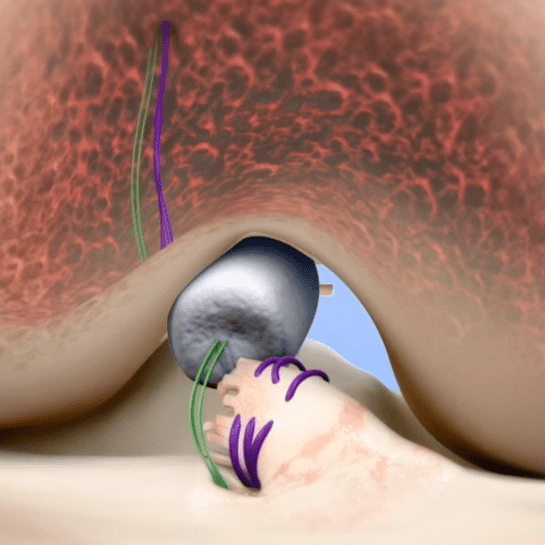
MIACH Orthopaedics is a privately-held company that develops implants for connective tissue repair. The company’s initial focus is on BEAR technology—a proprietary bio-engineered sponge used as a bridging scaffold to stimulate healing of a torn ACL. Unlike traditional ACL reconstruction, the BEAR procedure does not require taking grafts from healthy parts of the leg.
Preclinical studies, according to MIACH, suggest that knees treated with BEAR have a significantly lower rate of arthritis than those treated with traditional ACL reconstruction. The company has initiated an IDE clinical trial using ~100 human patients in order to generate sufficient data for a PMA.
BEAR technology was pioneered by Martha Murray, M.D., at the Boston Children’s Hospital Department of Orthopaedic Surgery. Initial research funding was provided by the NFL Players Association, Boston Children’s Hospital and the National Institutes of Health.
Source: MIACH Orthopaedics; image courtesy of MIACH Orthopaedics
MIACH Orthopaedics closed a US $22.5MM round of Series A financing. Funds will support accelerated manufacturing and continued clinical trials for the company’s first implant, Bridge-Enhanced® ACL Repair (BEAR®), designed to stimulate healing of torn anterior cruciate ligaments (ACL).
The round...
 | |||||
MIACH Orthopaedics closed a US $22.5MM round of Series A financing. Funds will support accelerated manufacturing and continued clinical trials for the company’s first implant, Bridge-Enhanced® ACL Repair (BEAR®), designed to stimulate healing of torn anterior cruciate ligaments (ACL).
The round was co-led by Amzak Health Investors LLC and DSM Venturing, with participation from the NFL Players Association and additional investors.
MIACH Orthopaedics is a privately-held company that develops implants for connective tissue repair. The company’s initial focus is on BEAR technology—a proprietary bio-engineered sponge used as a bridging scaffold to stimulate healing of a torn ACL. Unlike traditional ACL reconstruction, the BEAR procedure does not require taking grafts from healthy parts of the leg.
Preclinical studies, according to MIACH, suggest that knees treated with BEAR have a significantly lower rate of arthritis than those treated with traditional ACL reconstruction. The company has initiated an IDE clinical trial using ~100 human patients in order to generate sufficient data for a PMA.
BEAR technology was pioneered by Martha Murray, M.D., at the Boston Children’s Hospital Department of Orthopaedic Surgery. Initial research funding was provided by the NFL Players Association, Boston Children’s Hospital and the National Institutes of Health.
Source: MIACH Orthopaedics; image courtesy of MIACH Orthopaedics

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.