
 Copy to clipboard
Copy to clipboard 
MedShape announced that the U.S. Centers for Medicare & Medicaid Services has issued a new ICD-10-PCS code for a sustained compression internal fixation device in both lower and upper extremity joint fusion. The company’s DynaNail® TTC Fusion and DynaNail Mini® Fusion systems are the only devices approved for use under the new code.
Previously, no ICD-10-PCS code existed to specifically describe fusion procedures using internal fixation devices that could sustain compression post-surgery.
This new classification will enable healthcare providers and payers to selectively track the utilization, outcomes and healthcare costs associated with this class of NiTiNOL compression technology.
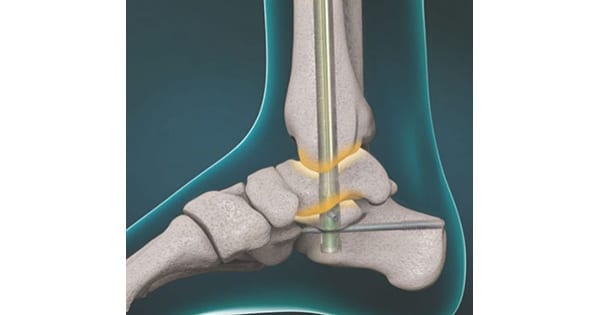
Unlike traditional static devices, DynaNail and DynaNail Mini offer sustained compression through a NiTiNOL Compressive Element that responds to bone resorption and other changes during bone healing. Unlike static devices, these provide immediate dynamization and have shown to effectively load-share with the surrounding bones.
MedShape announced that the U.S. Centers for Medicare & Medicaid Services has issued a new ICD-10-PCS code for a sustained compression internal fixation device in both lower and upper extremity joint fusion. The company's DynaNail® TTC Fusion and DynaNail Mini® Fusion systems are the only devices approved for use under the new code.
...
MedShape announced that the U.S. Centers for Medicare & Medicaid Services has issued a new ICD-10-PCS code for a sustained compression internal fixation device in both lower and upper extremity joint fusion. The company’s DynaNail® TTC Fusion and DynaNail Mini® Fusion systems are the only devices approved for use under the new code.
Previously, no ICD-10-PCS code existed to specifically describe fusion procedures using internal fixation devices that could sustain compression post-surgery.
This new classification will enable healthcare providers and payers to selectively track the utilization, outcomes and healthcare costs associated with this class of NiTiNOL compression technology.
Unlike traditional static devices, DynaNail and DynaNail Mini offer sustained compression through a NiTiNOL Compressive Element that responds to bone resorption and other changes during bone healing. Unlike static devices, these provide immediate dynamization and have shown to effectively load-share with the surrounding bones.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







