Copy to clipboard
Copy to clipboard 
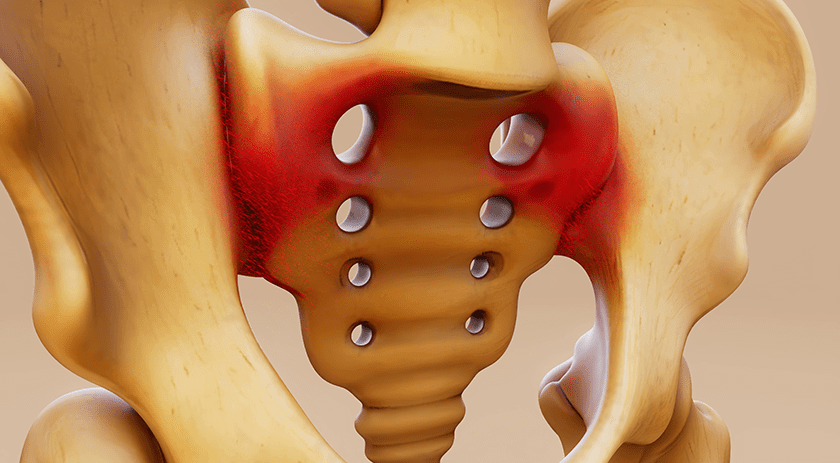
Medicrea gained FDA 510(k) clearance to integrate a complete database of implants into its UNiD HUB™ surgical planning software, to directly simulate and select implants prior to a procedure.
This allows every implant to be planned prior to surgery, leaving staff to focus on inter-op care rather than excess inventory. It also yields savings in cost and human effort required to complete reprocessing and sterilization of a spine system, due to a reduction in the number of trays needed.
Referring to beta phase use of the database with UNiD HUB, Denys Sournac, President and CEO, said, “[When a set is] loaned for a specific surgery, less than 10% of the inventory shipped is implanted in the patient. By pre-operatively measuring the patient’s pedicle diameter and length, we were able to reduce by 70% the amount of inventory being shipped. Similarly, in hospitals where inventory is consigned, we were able to optimize the number of implants and observed a reduction of inventory of 80% during the beta phase.”
Further benefits of the database integration will support the ability to perform comprehensive patient-specific preoperative planning that can more completely link implant selection with post-op analyses, and increase the robustness and efficiency of Medicrea’s proprietary predictive model.
Source: Medicrea
Medicrea gained FDA 510(k) clearance to integrate a complete database of implants into its UNiD HUB™ surgical planning software, to directly simulate and select implants prior to a procedure.
This allows every implant to be planned prior to surgery, leaving staff to focus on inter-op care rather than excess inventory. It also yields savings in...
Medicrea gained FDA 510(k) clearance to integrate a complete database of implants into its UNiD HUB™ surgical planning software, to directly simulate and select implants prior to a procedure.
This allows every implant to be planned prior to surgery, leaving staff to focus on inter-op care rather than excess inventory. It also yields savings in cost and human effort required to complete reprocessing and sterilization of a spine system, due to a reduction in the number of trays needed.
Referring to beta phase use of the database with UNiD HUB, Denys Sournac, President and CEO, said, “[When a set is] loaned for a specific surgery, less than 10% of the inventory shipped is implanted in the patient. By pre-operatively measuring the patient’s pedicle diameter and length, we were able to reduce by 70% the amount of inventory being shipped. Similarly, in hospitals where inventory is consigned, we were able to optimize the number of implants and observed a reduction of inventory of 80% during the beta phase.”
Further benefits of the database integration will support the ability to perform comprehensive patient-specific preoperative planning that can more completely link implant selection with post-op analyses, and increase the robustness and efficiency of Medicrea’s proprietary predictive model.
Source: Medicrea

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.