
 Copy to clipboard
Copy to clipboard 
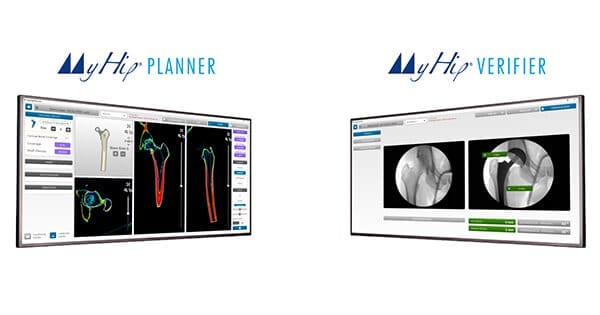
Medacta was granted CE Mark approval and FDA 510(k) clearance to market the MyHip® Planner and MyHip® Verifier, two personalized solutions intended for use in primary total hip replacement for 3D pre-operative planning and intra-operative verification.
Designed to predict and minimize surgical complexity, as well as to improve overall surgical outcomes and patient satisfaction, these applications can be used as stand-alone tools or together, and are intended to deliver a personalized approach to total hip arthroplasty.
MyHip Planner allows the user to create a patient-specific pre-operative 3D plan, tailoring implant choice and position to the patient’s anatomy, hip joint biomechanics, and functional needs.
MyHip Verifier uses intra-operative fluoroscopic images to verify patient-specific implant positioning.
The tools are part of the MySolutions Platform, Medacta’s personalized medicine offering, which also includes MyHip 3D printed patient-specific guides, POP® – Patient Optimized Pathway and MyClinical Data.
CEO Francesco Siccardi said, “Our goal is to bring value at every step throughout the entire treatment journey, while supporting surgeons to improve patient outcomes and maximizing value for the healthcare system. Our ecosystem of personalized and highly technological solutions will be soon enhanced with our new NextAR augmented reality-based surgical platform, which we have recently launched for total knee arthroplasty, and which we are working to integrate in our hip portfolio.”
Medacta was granted CE Mark approval and FDA 510(k) clearance to market the MyHip® Planner and MyHip® Verifier, two personalized solutions intended for use in primary total hip replacement for 3D pre-operative planning and intra-operative verification.
Designed to predict and minimize surgical complexity, as well as to improve overall...
Medacta was granted CE Mark approval and FDA 510(k) clearance to market the MyHip® Planner and MyHip® Verifier, two personalized solutions intended for use in primary total hip replacement for 3D pre-operative planning and intra-operative verification.
Designed to predict and minimize surgical complexity, as well as to improve overall surgical outcomes and patient satisfaction, these applications can be used as stand-alone tools or together, and are intended to deliver a personalized approach to total hip arthroplasty.
MyHip Planner allows the user to create a patient-specific pre-operative 3D plan, tailoring implant choice and position to the patient’s anatomy, hip joint biomechanics, and functional needs.
MyHip Verifier uses intra-operative fluoroscopic images to verify patient-specific implant positioning.
The tools are part of the MySolutions Platform, Medacta’s personalized medicine offering, which also includes MyHip 3D printed patient-specific guides, POP® – Patient Optimized Pathway and MyClinical Data.
CEO Francesco Siccardi said, “Our goal is to bring value at every step throughout the entire treatment journey, while supporting surgeons to improve patient outcomes and maximizing value for the healthcare system. Our ecosystem of personalized and highly technological solutions will be soon enhanced with our new NextAR augmented reality-based surgical platform, which we have recently launched for total knee arthroplasty, and which we are working to integrate in our hip portfolio.”

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







