
 Copy to clipboard
Copy to clipboard 
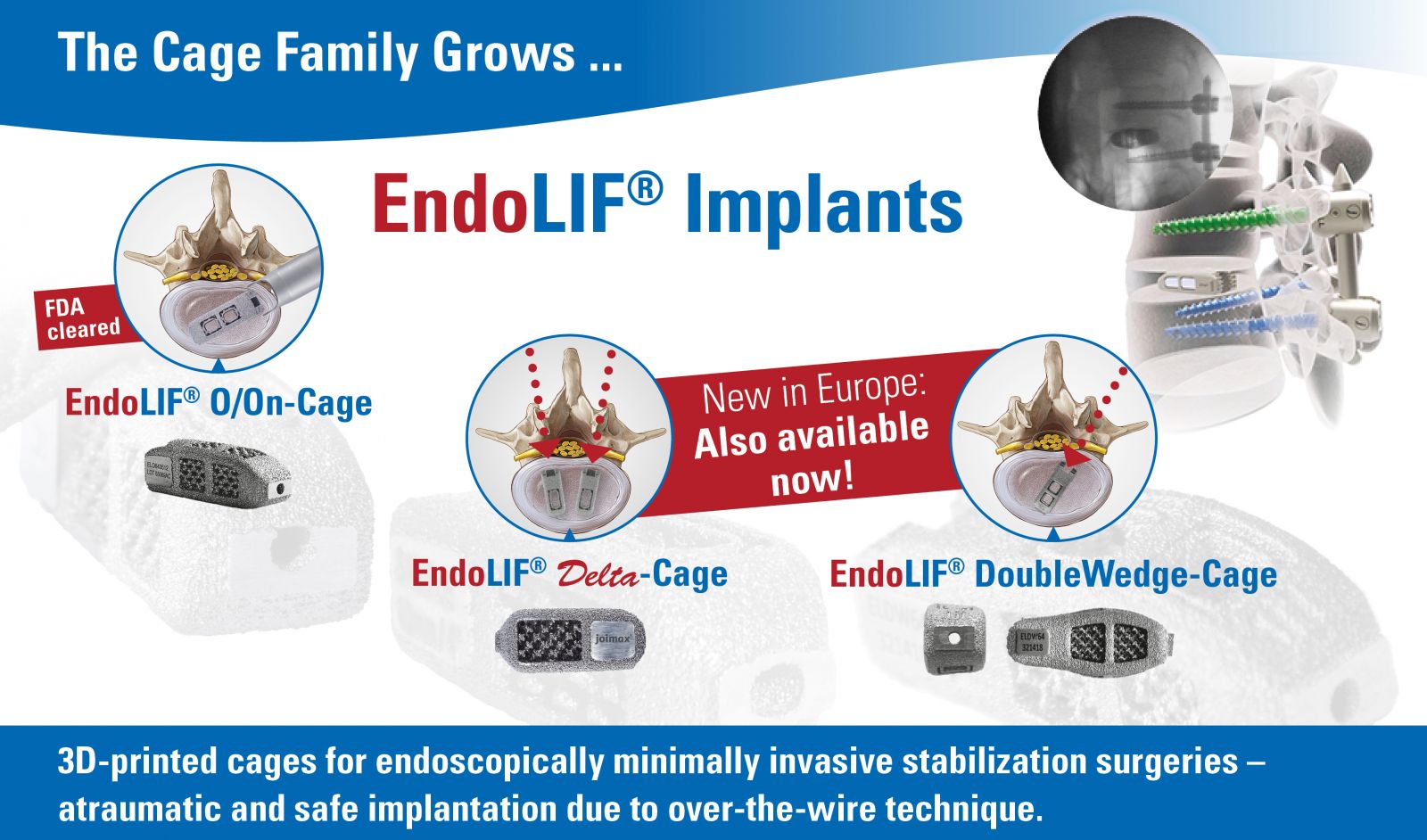
joimax commenced full launch of the complete line of EndoLIF® devices for minimally invasive lumbar interbody fusion.
The portfolio comprises three titanium alloy-based devices: EndoLIF® O/On-Cage, EndoLIF® Delta Cage and the latest, EndoLIF® Double Wedge Cage. All are FDA-cleared and CE Mark approved.
EndoLIF products are manufactured using the Electron Beam Melting 3D-printing process, which yields an open diamond cell structure. The porous surface is designed to encourage bone ingrowth and stability. One instrument set supports all three devices.
joimax markets EndoLIF for spinal stabilization and the Endoscopic Surgical Systems TESSYS® (transforaminal), iLESSYS® (interlaminar) and CESSYS® (cervical) for decompression procedures, MultiZYTE® for facet and sacroiliac joint pain treatment and EndoLIF® and Percusys® for minimally invasive endoscopically assisted stabilizations.
Source: joimax
joimax commenced full launch of the complete line of EndoLIF® devices for minimally invasive lumbar interbody fusion.
The portfolio comprises three titanium alloy-based devices: EndoLIF® O/On-Cage, EndoLIF® Delta Cage and the latest, EndoLIF® Double Wedge Cage. All are FDA-cleared and CE Mark approved.
EndoLIF products are manufactured...
joimax commenced full launch of the complete line of EndoLIF® devices for minimally invasive lumbar interbody fusion.
The portfolio comprises three titanium alloy-based devices: EndoLIF® O/On-Cage, EndoLIF® Delta Cage and the latest, EndoLIF® Double Wedge Cage. All are FDA-cleared and CE Mark approved.
EndoLIF products are manufactured using the Electron Beam Melting 3D-printing process, which yields an open diamond cell structure. The porous surface is designed to encourage bone ingrowth and stability. One instrument set supports all three devices.
joimax markets EndoLIF for spinal stabilization and the Endoscopic Surgical Systems TESSYS® (transforaminal), iLESSYS® (interlaminar) and CESSYS® (cervical) for decompression procedures, MultiZYTE® for facet and sacroiliac joint pain treatment and EndoLIF® and Percusys® for minimally invasive endoscopically assisted stabilizations.

Source: joimax

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







