Copy to clipboard
Copy to clipboard 
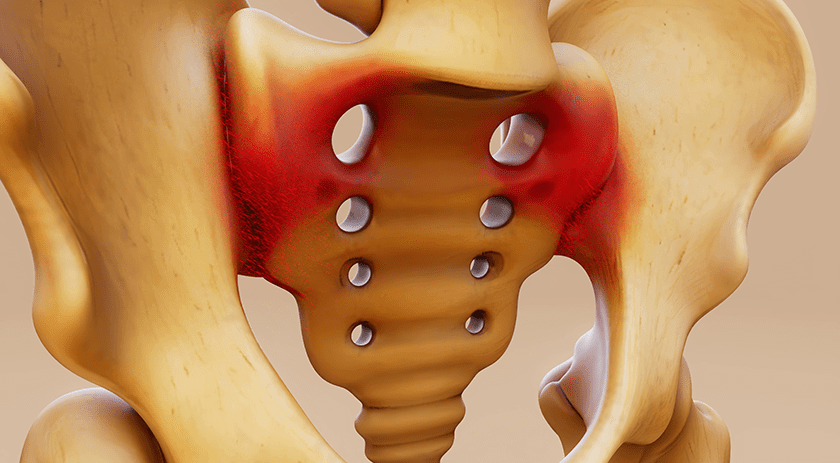
Intellirod Spine gained FDA de novo clearance for the LOADPRO® disposable spinal rod strain sensor. The device clamps to a 5.5mm rod to monitor rod strain during kyphotic correction. This initial approval is for use of LOADPRO with Xtant Medical’s FORTEX pedicle screw; first launches of the product are slated for Ohio and Kentucky. Expanded commercial launch sites and clearance for use with other pedice screw systems are in development.
This reportedly represents the first de novo clearance for a spine product.
“Post-market studies will add greater understanding of strains induced on the rods, the effect of different surgeon techniques on rod strain, and how reproducible procedures are relative to rod strain,” says Dr. Rolando M. Puno, spine surgeon and co-founder of Intellirod. “Ultimately, we want to understand how our loading of the implants during surgery can affect the outcomes. Implant-related complications in long construct kyphotic deformities can be as high as 20%. LOADPRO gives us the tool to do these investigations and to learn the implications of our techniques.”
Source: Intellirod Spine
Intellirod Spine gained FDA de novo clearance for the LOADPRO® disposable spinal rod strain sensor. The device clamps to a 5.5mm rod to monitor rod strain during kyphotic correction. This initial approval is for use of LOADPRO with Xtant Medical's FORTEX pedicle screw; first launches of the product are slated for Ohio and Kentucky. Expanded...
Intellirod Spine gained FDA de novo clearance for the LOADPRO® disposable spinal rod strain sensor. The device clamps to a 5.5mm rod to monitor rod strain during kyphotic correction. This initial approval is for use of LOADPRO with Xtant Medical’s FORTEX pedicle screw; first launches of the product are slated for Ohio and Kentucky. Expanded commercial launch sites and clearance for use with other pedice screw systems are in development.
This reportedly represents the first de novo clearance for a spine product.
“Post-market studies will add greater understanding of strains induced on the rods, the effect of different surgeon techniques on rod strain, and how reproducible procedures are relative to rod strain,” says Dr. Rolando M. Puno, spine surgeon and co-founder of Intellirod. “Ultimately, we want to understand how our loading of the implants during surgery can affect the outcomes. Implant-related complications in long construct kyphotic deformities can be as high as 20%. LOADPRO gives us the tool to do these investigations and to learn the implications of our techniques.”
Source: Intellirod Spine

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.