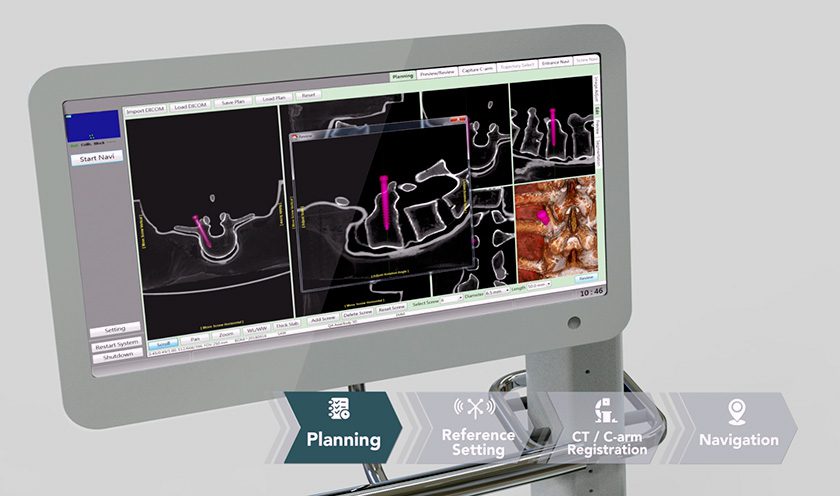
Innovasis received FDA 510(k) clearance to market the PEEK-OPTIMA® HA Enhanced LxHA™ device for lateral interbody fusion. Full launch is slated for 3Q19.
LxHA will complement a new next generation retractor system launched to be launched by the end of the year.
The interbody device features a bullet-shaped nose, large central graft window and pyramid-shaped teeth to help resist migration. It is intended for use with internal supplemental spinal fixation, such as Innovasis Excella®.
Source: Innovasis, Inc.
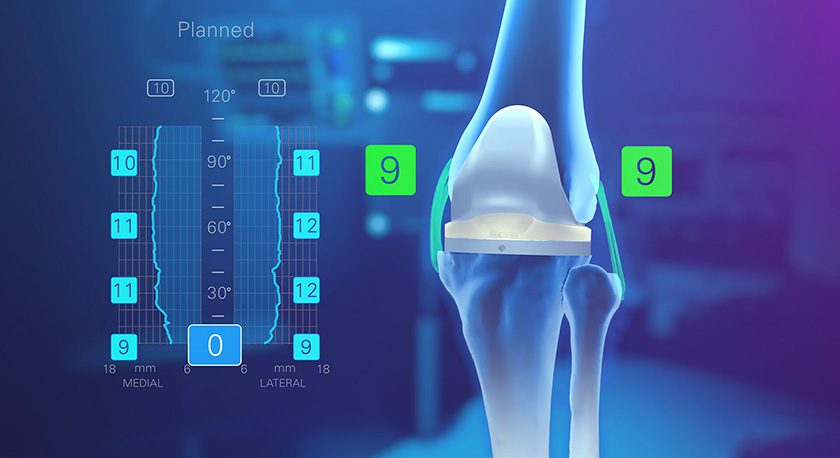
Innovasis received FDA 510(k) clearance to market the PEEK-OPTIMA® HA Enhanced LxHA™ device for lateral interbody fusion. Full launch is slated for 3Q19.
LxHA will complement a new next generation retractor system launched to be launched by the end of the year.
The interbody device features a bullet-shaped nose, large central graft window...
Innovasis received FDA 510(k) clearance to market the PEEK-OPTIMA® HA Enhanced LxHA™ device for lateral interbody fusion. Full launch is slated for 3Q19.
LxHA will complement a new next generation retractor system launched to be launched by the end of the year.
The interbody device features a bullet-shaped nose, large central graft window and pyramid-shaped teeth to help resist migration. It is intended for use with internal supplemental spinal fixation, such as Innovasis Excella®.
Source: Innovasis, Inc.


You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.