
 Copy to clipboard
Copy to clipboard 
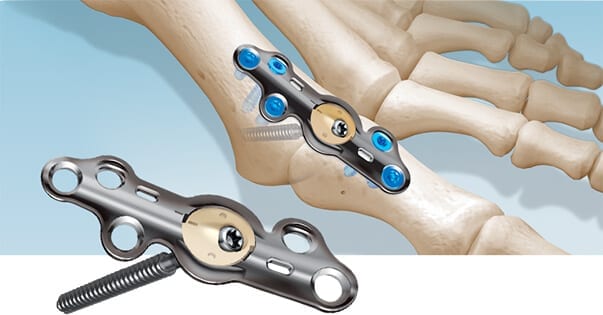
In2Bones USA received FDA 510(k) clearance to market CoLink® View Bone Plates for the CoLink forefoot bone plating system.
CoLink View plates are made from titanium and PEEK, and incorporate an x-ray transparent PEEK hub that essentially enables a view of bone as it heals. Further, the PEEK material mimics bone’s own elasticity.
In2Bones’ other PEEK-based products include the NeoView® Distal Radius Plate and the PitStop™ subtalar implant. Other PEEK-based implants are slated for FDA clearance in 2018.
Source: In2Bones USA, LLC
In2Bones USA received FDA 510(k) clearance to market CoLink® View Bone Plates for the CoLink forefoot bone plating system.
CoLink View plates are made from titanium and PEEK, and incorporate an x-ray transparent PEEK hub that essentially enables a view of bone as it heals. Further, the PEEK material mimics bone's own elasticity.
In2Bones'...
In2Bones USA received FDA 510(k) clearance to market CoLink® View Bone Plates for the CoLink forefoot bone plating system.
CoLink View plates are made from titanium and PEEK, and incorporate an x-ray transparent PEEK hub that essentially enables a view of bone as it heals. Further, the PEEK material mimics bone’s own elasticity.
In2Bones’ other PEEK-based products include the NeoView® Distal Radius Plate and the PitStop™ subtalar implant. Other PEEK-based implants are slated for FDA clearance in 2018.
Source: In2Bones USA, LLC

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







