Copy to clipboard
Copy to clipboard 
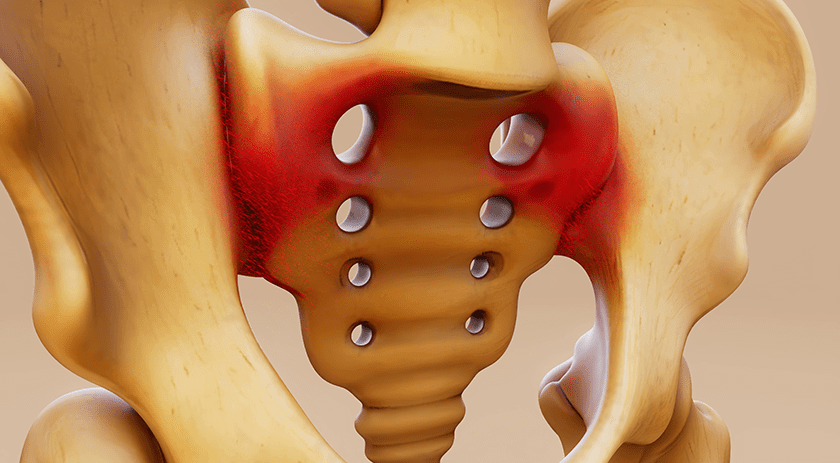
Implanet received FDA 510(k) clearance to market JAZZ Cap®, its system to secure spinal screws in poor quality bone for vertebral fusion procedures.
JAZZ Cap was developed for the treatment of degenerative conditions in adult patients. The clearance plays a key role in in the private label distribution agreement signed with SeaSpine earlier this year, whereby SeaSpine has exclusive rights to sell JAZZ products, including JAZZ Cap, under its own brand name.
This is the company’s tenth 510(k) for components in the JAZZ portfolio, and it follows the product’s CE Mark Approval that was secured in 4Q18.
Sources: Implanet; ORTHOWORLD Inc.
Implanet received FDA 510(k) clearance to market JAZZ Cap®, its system to secure spinal screws in poor quality bone for vertebral fusion procedures.
JAZZ Cap was developed for the treatment of degenerative conditions in adult patients. The clearance plays a key role in in the private label distribution agreement signed with SeaSpine earlier...
Implanet received FDA 510(k) clearance to market JAZZ Cap®, its system to secure spinal screws in poor quality bone for vertebral fusion procedures.
JAZZ Cap was developed for the treatment of degenerative conditions in adult patients. The clearance plays a key role in in the private label distribution agreement signed with SeaSpine earlier this year, whereby SeaSpine has exclusive rights to sell JAZZ products, including JAZZ Cap, under its own brand name.
This is the company’s tenth 510(k) for components in the JAZZ portfolio, and it follows the product’s CE Mark Approval that was secured in 4Q18.
Sources: Implanet; ORTHOWORLD Inc.

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.