
 Copy to clipboard
Copy to clipboard 
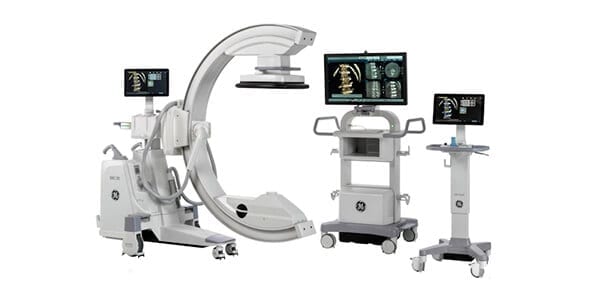
GE Healthcare was granted FDA 510(k) clearance to market OEC 3D, a new surgical imaging system capable of 3D and 2D imaging.
This new system combines the benefits and familiarity of 2D imaging with greater efficiency to increase access and usability to 3D in orthopedic and spine procedures.
OEC 3D provides a large 19cm x 19cm x 19cm Field of View with high resolution and detail. An advanced reconstruction engine quickly presents images on an intuitive Volume Viewer with tools and analysis for surgical assessment. OEC 3D can interface with intraoperative technologies such as navigation and robotics.
For improved asset utilization, OEC 3D includes the recognizable 2D imaging experience of OEC C-arms for imaging versatility from general surgery to interventional cardiovascular procedures, delivering a familiar user experience with the added benefit to quickly capture a 3D scan.
“We’re thrilled to introduce OEC 3D to clinicians who want amazing 3D volumetric images quickly during intraoperative procedures,” said Gustavo Perez Fernandez, president and CEO of GE Healthcare Image Guided Therapies. “Built on the successful OEC Elite C-arm platform, the familiar performance and functionality of the OEC 3D C-arm will make 3D imaging routine for complex spine and joint replacement procedures.”
GE Healthcare was granted FDA 510(k) clearance to market OEC 3D, a new surgical imaging system capable of 3D and 2D imaging.
This new system combines the benefits and familiarity of 2D imaging with greater efficiency to increase access and usability to 3D in orthopedic and spine procedures.
OEC 3D provides a large 19cm x 19cm x 19cm...
GE Healthcare was granted FDA 510(k) clearance to market OEC 3D, a new surgical imaging system capable of 3D and 2D imaging.
This new system combines the benefits and familiarity of 2D imaging with greater efficiency to increase access and usability to 3D in orthopedic and spine procedures.
OEC 3D provides a large 19cm x 19cm x 19cm Field of View with high resolution and detail. An advanced reconstruction engine quickly presents images on an intuitive Volume Viewer with tools and analysis for surgical assessment. OEC 3D can interface with intraoperative technologies such as navigation and robotics.
For improved asset utilization, OEC 3D includes the recognizable 2D imaging experience of OEC C-arms for imaging versatility from general surgery to interventional cardiovascular procedures, delivering a familiar user experience with the added benefit to quickly capture a 3D scan.
“We’re thrilled to introduce OEC 3D to clinicians who want amazing 3D volumetric images quickly during intraoperative procedures,” said Gustavo Perez Fernandez, president and CEO of GE Healthcare Image Guided Therapies. “Built on the successful OEC Elite C-arm platform, the familiar performance and functionality of the OEC 3D C-arm will make 3D imaging routine for complex spine and joint replacement procedures.”

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







