
 Copy to clipboard
Copy to clipboard 
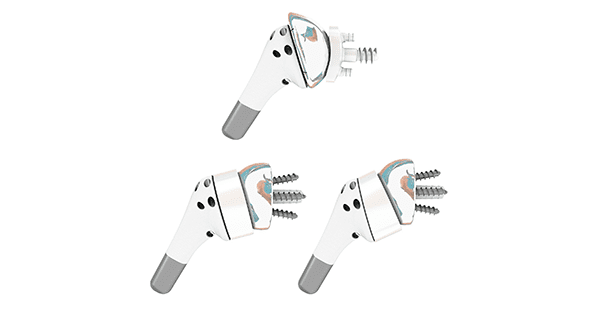
FX Solutions received FDA 510(k) clearance for their new FX V135® shoulder replacement platform which includes a new-to-market mini stem humeral component that is set at 135° for both the anatomical and reverse configurations.
With FX’s net shape molded humeral cups, the reverse construct also allows surgeons the option to use an asymmetric humeral cup to change the inclination to 145° through the humeral cup component.
The FX V135 anatomical configuration features a taper component that will allow for 360° of eccentricity providing for a dual eccentricity offering between the taper and the humeral head components. New with this system, FX is launching humeral head components with variable head heights to allow surgeons more flexibility to best match the patients’ anatomy.
The FX V135 is a 70mm humeral stem that is proximally coated with hydroxyapatite featuring suture holes proximally for soft tissue repair, unique-to-market net shape molded humeral cups, Titanium Nitride coated and uncoated Cobalt Chrome humeral heads and glenospheres, and 12 different baseplate options in the reverse construct. The humeral stem inclination is at 135° for the anatomical construct and 135° or 145° for the reverse construct.
Source: FX Solutions
FX Solutions received FDA 510(k) clearance for their new FX V135® shoulder replacement platform which includes a new-to-market mini stem humeral component that is set at 135° for both the anatomical and reverse configurations.
With FX's net shape molded humeral cups, the reverse construct also allows surgeons the option to use an asymmetric...
FX Solutions received FDA 510(k) clearance for their new FX V135® shoulder replacement platform which includes a new-to-market mini stem humeral component that is set at 135° for both the anatomical and reverse configurations.
With FX’s net shape molded humeral cups, the reverse construct also allows surgeons the option to use an asymmetric humeral cup to change the inclination to 145° through the humeral cup component.
The FX V135 anatomical configuration features a taper component that will allow for 360° of eccentricity providing for a dual eccentricity offering between the taper and the humeral head components. New with this system, FX is launching humeral head components with variable head heights to allow surgeons more flexibility to best match the patients’ anatomy.
The FX V135 is a 70mm humeral stem that is proximally coated with hydroxyapatite featuring suture holes proximally for soft tissue repair, unique-to-market net shape molded humeral cups, Titanium Nitride coated and uncoated Cobalt Chrome humeral heads and glenospheres, and 12 different baseplate options in the reverse construct. The humeral stem inclination is at 135° for the anatomical construct and 135° or 145° for the reverse construct.
Source: FX Solutions

You are out of free articles for this month
Subscribe as a Guest for $0 and unlock a total of 5 articles per month.
You are out of five articles for this month
Subscribe as an Executive Member for access to unlimited articles, THE ORTHOPAEDIC INDUSTRY ANNUAL REPORT and more.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







