
 Copy to clipboard
Copy to clipboard 
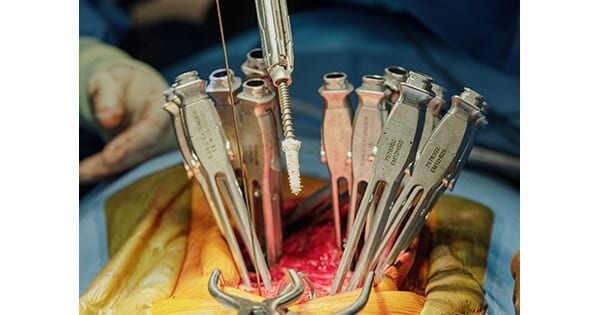
TheraCell completed the first three spine surgical cases using the world’s first-ever demineralized bone fiber screw anchor. Fiber Anchors secured screws at three levels, and TheraFuze DBF® Fiber Wraps™ were used with 10cc of TheraFuze DBF FiberForm™+ for a two-level fusion.
The system acts like a drywall anchor, providing immediate improvement in screw fixation in primary and revision cases.
The TheraFuze DBF Fiber Anchor is made from TheraCell’s proprietary demineralized cortical bone fibers. Testing demonstrated it more than doubles the pullout force versus screws without the Fiber Anchor. The TheraFuze DBF Fiber Wrap contains demineralized bone fibers formed into a 1mm thick sheet with TheraCell’s FiberLok™ technology. TheraFuze DBF FiberForm+ is a moldable demineralized excipient-free putty for use in orthopedic and spine surgery that also includes mineralized cortical chips. Cortical chips provide additional compression resistance and radiodensity for visualization.
Curt Cooper, TheraCell’s Chief Business Development Officer, said, “The TheraFuze DBF Fiber Anchor offers a cost-effective solution for revision cases or where the surgeon “feels” the bone quality is compromised as it provides significant improved initial in vivo fixation. In our pre-clinical studies, the Fiber Anchor demonstrated that it promotes new bone formation in apposition to the screw with interdigitation of remodeled bone into the threads surrounded by native bone. Proud to develop a highly differentiated clinical solution, we believe the Fiber Anchor provides surgeons a novel, safe alternative to Polymethyl Methacrylate (PMMA).”
TheraCell completed the first three spine surgical cases using the world's first-ever demineralized bone fiber screw anchor. Fiber Anchors secured screws at three levels, and TheraFuze DBF® Fiber Wraps™ were used with 10cc of TheraFuze DBF FiberForm™+ for a two-level fusion.
The system acts like a drywall anchor, providing immediate...
TheraCell completed the first three spine surgical cases using the world’s first-ever demineralized bone fiber screw anchor. Fiber Anchors secured screws at three levels, and TheraFuze DBF® Fiber Wraps™ were used with 10cc of TheraFuze DBF FiberForm™+ for a two-level fusion.
The system acts like a drywall anchor, providing immediate improvement in screw fixation in primary and revision cases.
The TheraFuze DBF Fiber Anchor is made from TheraCell’s proprietary demineralized cortical bone fibers. Testing demonstrated it more than doubles the pullout force versus screws without the Fiber Anchor. The TheraFuze DBF Fiber Wrap contains demineralized bone fibers formed into a 1mm thick sheet with TheraCell’s FiberLok™ technology. TheraFuze DBF FiberForm+ is a moldable demineralized excipient-free putty for use in orthopedic and spine surgery that also includes mineralized cortical chips. Cortical chips provide additional compression resistance and radiodensity for visualization.
Curt Cooper, TheraCell’s Chief Business Development Officer, said, “The TheraFuze DBF Fiber Anchor offers a cost-effective solution for revision cases or where the surgeon “feels” the bone quality is compromised as it provides significant improved initial in vivo fixation. In our pre-clinical studies, the Fiber Anchor demonstrated that it promotes new bone formation in apposition to the screw with interdigitation of remodeled bone into the threads surrounded by native bone. Proud to develop a highly differentiated clinical solution, we believe the Fiber Anchor provides surgeons a novel, safe alternative to Polymethyl Methacrylate (PMMA).”

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







