
 Copy to clipboard
Copy to clipboard 
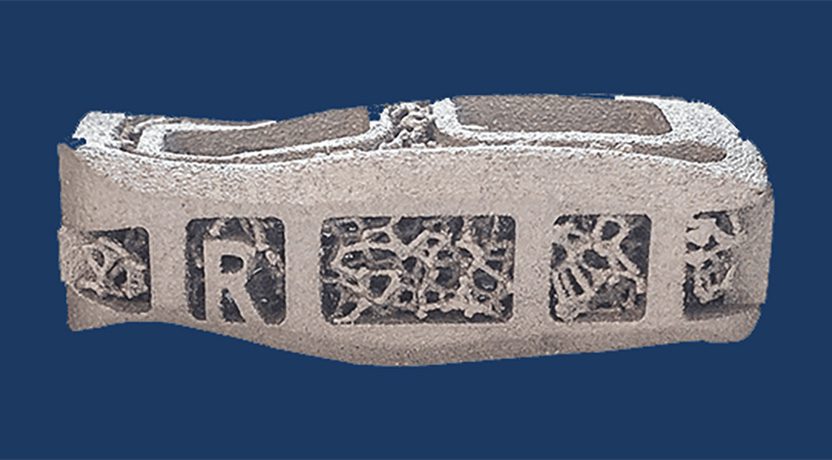
FDA granted Breakthrough Device designation to Carlsmed for its aprevo technology for the treatment of cervical spine disease. This is Carlsmed’s second Breakthrough Device designation, complementing the company’s family of lumbar patient-specific interbody fusion devices for anterior, lateral and transforaminal approaches.
Carlsmed’s digital technology platform creates unique fusion devices for each patient. The bespoke aprevo devices are 3D printed, sterile packed and delivered directly to the hospital for surgery.
Carlsmed’s aprevo device for the treatment of cervical spine disease is slated for U.S. launch in 2025.
Source: Carlsmed
FDA granted Breakthrough Device designation to Carlsmed for its aprevo technology for the treatment of cervical spine disease. This is Carlsmed’s second Breakthrough Device designation, complementing the company’s family of lumbar patient-specific interbody fusion devices for anterior, lateral and transforaminal approaches.
Carlsmed’s...
FDA granted Breakthrough Device designation to Carlsmed for its aprevo technology for the treatment of cervical spine disease. This is Carlsmed’s second Breakthrough Device designation, complementing the company’s family of lumbar patient-specific interbody fusion devices for anterior, lateral and transforaminal approaches.
Carlsmed’s digital technology platform creates unique fusion devices for each patient. The bespoke aprevo devices are 3D printed, sterile packed and delivered directly to the hospital for surgery.
Carlsmed’s aprevo device for the treatment of cervical spine disease is slated for U.S. launch in 2025.
Source: Carlsmed

You’ve reached your limit.
We’re glad you’re finding value in our content — and we’d love for you to keep going.
Subscribe now for unlimited access to orthopedic business intelligence.
JV
Julie Vetalice is ORTHOWORLD's Editorial Assistant. She has covered the orthopedic industry for over 20 years, having joined the company in 1999.







